RESEARCH NOTE
Jeff A. Alvarez1* and Barbara J. Alvarez2
1 The Wildlife Project, P.O. Box 188888, Sacramento, CA 95818, USA
2 774 5th Avenue, Sacramento, CA 95818, USA
*Corresponding Author: jeff@thewildlifeproject.com
Published 1 July 2022 • http://www.doi.org/10.51492/cfwj.108.8
Key words: allopatric, breeding, control, disjunct populations, established, expansion, invasive, Pomacea canaliculata, range
A recent focus on globalization as the origin of a surge in invasive species translocations has been reported (Perrings et al. 2005; Meyerson and Mooney 2007; Hulme 2009). The general trend to the open transit of goods and services worldwide has caused some researchers to speculate or even predict that invasive species will become a greater problem in the future (Lodge et al. 2006; McNeely 2006; Sutherland et al. 2008). Unfortunately, invasive species have been considered the cause of or a contributing factor in the extinction of many species from several classes of wildlife (Clavero and Garcia-Berthou 2005). In particular, aquatic systems appear to be the most severely impacted ecosystems (Moyle 1999), with nearly all freshwater catchments in North America containing at least one non-native species (Gido and Brown 1999). Nevertheless, Williamson (1996) suggested that non-native species invasions are a process that includes different stages, including transport, establishment, and spread. Occasionally this process is not successful but can unfortunately be repeated several or many times before establishment (Moyle 1999).
In California, numerous invasive species have been translocated to this novel environment with a great number becoming established (Ingram 1959; Grosholz and Ruiz 1995; Moyle 1999; Bolch et al. 2021). In the aquatic environment, Moyle (1999) reported at least 49 fish species that have become established with an additional 38 that failed. Numerous invertebrate species have also been introduced into California, with many more remaining undetected or unreported (Ingram 1959; Cohen et al. 1995; Grosholz and Ruiz 1995; Cohen and Carlton 1997).
In the late 1990s, the channeled (= golden) apple snail (Pomacea canaliculata, which has been reported as one of the most negatively impactful invasive species on earth (Lowe et al. 2000), was found in California (Cerutti 1998; Howells et al. 2006). Initially, these snails were thought to be introduced for the purpose of food for humans. Subsequent introductions or reintroductions, however, may have been generated from the aquatic pet trade (Cowie et el. 2017). The origin of this species is South America, specifically, Argentina, Bolivia, Paraguay, Uruguay, and Brazil (Rawlings et al. 2007), where it likely occupied similar habitats to those found in portions of the United States. This species is reported to be a voracious consumer of aquatic vegetation and is expected to compete for resources with native mollusks and other invertebrates (Naylor 1996; Wood et al. 2005). They are also believed to be a vector for the rat lungworm (Angiostrongylus cantonensis), which can cause significant illness in humans (Tseng et al. 2011; Lv et al. 2009). In California, if the species were to spread, it would be considered a threat to rice crops (Naylor 1996; Yeo 2004; Global Invasive Species Database 2021).
Herein, the current distribution and putative spread of P. canaliculata in California is reported. New locations where the species appears to have formed breeding populations are also reported.
We collected data from personal observations, previously reported/published works, museum collections, and confirmed community science reports. For community science reports, observations were verified if each report was supported by a photo that the authors could confirm, and/or included a site visit and subsequent search and confirmation of the species presence. We then categorized all sources of observations within decadal periods (i.e., 1990s, 2000s, 2010s, and 2020–2021). Changes were noted in the reported locations of observations or known occupied sites to determine if populations were increasing (geographically) and if new unassociated populations were becoming established. We mapped these data and compared them within decadal spans, when possible.
It was determined that P. canaliculata was first reported in California in 1997 from Miramar Lake in San Diego County (Cerutti 1998). In the same year, two other locations were also reported at Norton Simon Museum, Los Angeles County, and within drainage canals associated with the Salton Sea, in Riverside County (Howells et al. 2006). Howells et al. (2006) also noted a fourth site in Fremont, Alameda County that was subsequently reported as eradicated (Fig. 1a and 1b). A single additional site was reported between 2000 and 2010; P. canaliculata was found in a pond at the Los Coyotes Country Club, Orange County (USGS, unpublished data; Fig. 1b). In the 2010s, five additional counties in which this species was reported, as well as continued reports (population persistence) at the original sites (i.e., counties) in which it was known (Los Angeles, Orange, Riverside, and San Diego Counties; Fig. 1c.) were recorded. In 2021, P. canaliculata was reported in three additional counties (Fig. 1d and Fig. 2.). As of summer 2021, all 10 counties with extant populations were made up of breeding populations (J. Alvarez, personal observation).
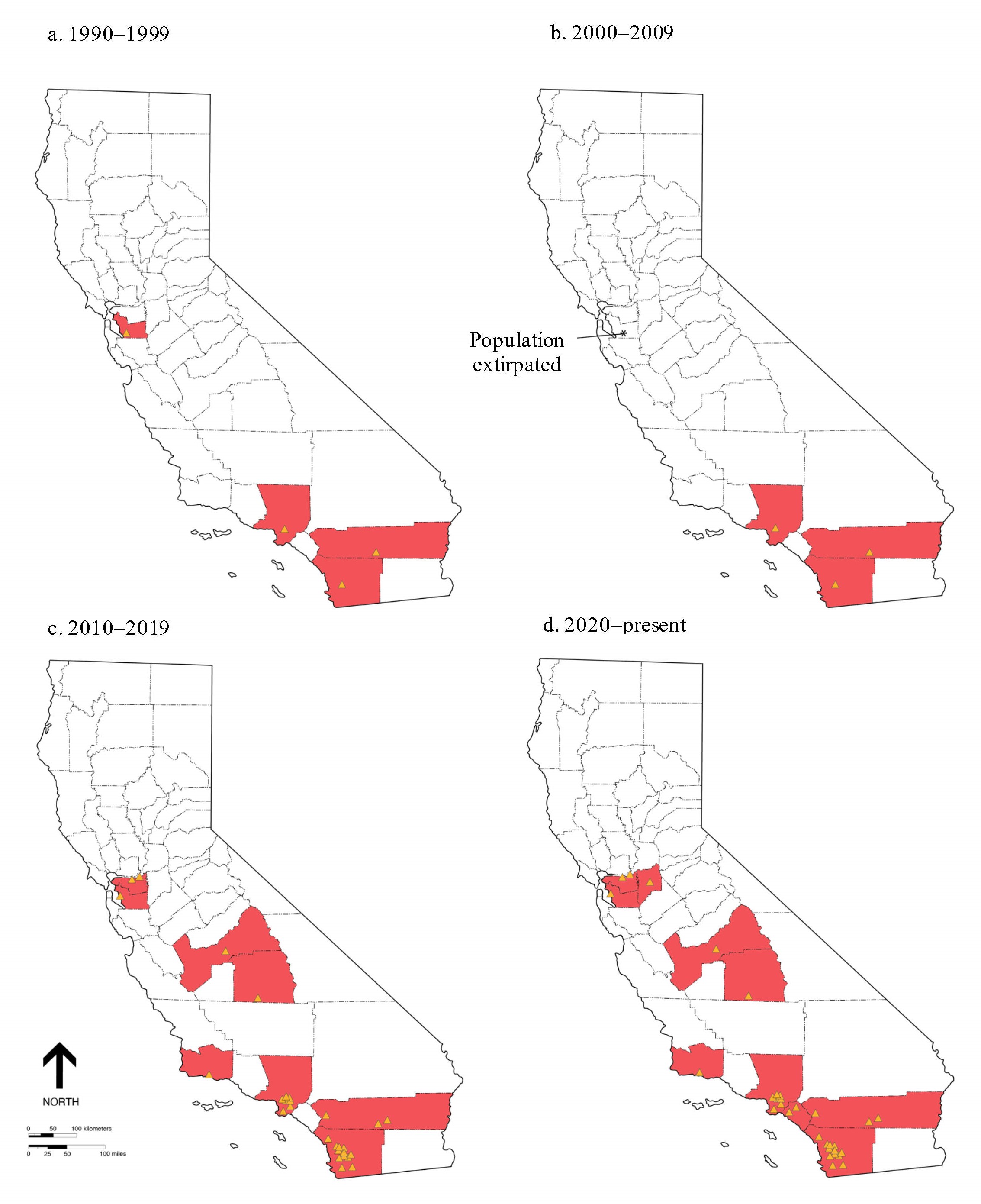

Given that the original populations were observed in 1997, it appears that they have been self-sustaining for at least 24 years. Given the current geographic extent and disjunct nature of these populations reported herein, it appears that every occupied county has had a separate introduction event. It is noted that only two of the known populations appear to be hydrologically connected. The population at Big Break (Antioch, CA) and Dutch Slough (Bethel Island, CA) in Contra Costa County are hydrologically connected through the Sacramento-San Joaquin River Delta. Although the authors did not observe population connectivity between Big Break and Dutch Slough directly, there remains a high likelihood that this is a very large extended population that reaches more than 6 km. Two disjunct populations that occur in Oasis and Mecca, Riverside County, California are hydrologically connected via the Salton Sea. However, the Salton Sea is reported to have a salinity of at least 44 ppt (Schroeder et al. 2002), while Wada and Matsukura (2009) suggested that a salinity level of approximately 4 ppt may be lethal to a “majority” of individual snails. It is, therefore, concluded that the Salton Sea is likely a dispersal barrier for this species from one drainage canal or river to another.
Observations by the authors indicate that 32 of the 33 currently extant populations are allopatric, and are part of an iterative range expansion, which appears to indicate that all populations were introduced separately and over time. The current distribution also indicates an invasion pathway that is associated with numerous anthropogenic releases over a geographic area that includes more than half the length of California. This type of introduced species invasion follows that postulated by Williamson (1996) who suggested that successful invasion is a process that happens overtime (see also: Moyle 1999).
Yeo (2004) and the Global Invasive Species Database (2021) both suggested that this species may greatly impact rice production if the species were to be introduced into areas adjacent to productive fields. With a potentially large population of P. canaliculata established in the Sacramento-San Joaquin River Delta, which is hydrologically connected to agricultural areas supporting rice farming to the north, it is likely that future management of this invasive species would include limiting or avoiding further encroachment of northern dispersal into sensitive agricultural areas.
Based on observations by the authors, introductions into suitable habitat can occur anywhere that a golf ball-sized mollusk could be carried. Speculation has been abundant for cold temperatures as limiting movement. Oya et al. (1987) and Mochida (1991), however reported that P. canaliculata could survive for up to 20 days at 0°C. This may be a limiting factor for this species in the Sierra Nevada Mountains but will not likely affect the spread of this snail in the northern Central Valley (i.e., Sacramento Valley) and lower elevations of the surrounding foothills. The current California climate, as well as potential future climate change may, in fact, facilitate occupation of millions of acres of California’s lakes, creeks, natural wetlands, mitigation areas, agricultural ditches, sewage treatment ponds, and other sites where this species may impact native flora and fauna (Schoenherr 2007; Joshi et al. 2017).
is clearly increasing in its overall geographic spread, nearly entirely through anthropogenic translocation. We suggest that regulations be put in place to limit or curtail any possession, breeding, transportation, or release of this species into any water way in California. Without vigorous control of new populations, and control over dispersal of extant populations, P. canaliculata will undoubtedly become established in more areas of California, increasing the threat of eventually becoming established in sensitive agricultural areas.
Acknowledgments
We wish to thank the California Department of Fish and Wildlife for their direction on the disposition of collected specimens. We also acknowledge the effort of M. Alvarez in surveying numerous golf course ponds for P. canaliculata, and for his comments on the manuscript. Two anonymous reviewers improved this manuscript substantially, and their efforts are greatly appreciated.
Literature Cited
- Bolch, E. A., E. L. Hestir, and S. Khanna. 2021. Performance and feasibility of drone-mounted imaging spectroscopy for invasive aquatic vegetation detection. Remote Sensing 13:582–601.
- Cerutti, R. 1998. An infestation of Pomacea canaliculata (Lamarck, 1804) in Lake Miramar, San Diego, California. The Festivus 30:25–27.
- Clavero, M., and E. García-Berthou. 2005. Invasive species are a leading cause of animal extinctions. Trends in Ecology and Evolution 20:110.
- Cohen, A. N., and J. T. Carlton. 1997. Transoceanic transport mechanisms: Introduction of the Chinese Mitten Crab, Eriocheir sinensis, to California. Pacific Science 51:1–11.
- Carcinus maenas in San Francisco Bay, California. Marine Biology 122:225–237.
- Gido, K. B., and J. H. Brown. 1999. Invasion of North American drainages by alien fish species. Freshwater Biology 42:387–399.
- Global Invasive Species Database. 2021. Species profile: Pomacea canaliculata. Available from: http://www.iucngisd.org/gisd/species.php?sc=135 (Accessed: 19 Sept 2021).
- Grosholz, E. D., and G. M. Ruiz. 1995. Spread and potential impact of the recently introduced European green crab, Carcinus maenas, in central California. Marine Biology 122: 239–247.
- Howells, R. G., L. E. Burlakova, A. Y. Karatayev, R. K. Marfurt, and R. L. Burks. 2006. Native and introduced Ampullariidae in North America: History, status, and ecology. Pages 1–40 in R. C. Joshi, and L. S. Sebastian, editors. Global Advances in Ecology and Management of Golden Apple Snails. Philippine Rice Research Institute, Nueva Ecija, Philippines.
- Hulme, P. E. 2009. Trade, transport, and trouble: Managing invasive species pathways in an era of globalization. Journal of Applied Ecology 46:10–18.
- Ingram, W. M. 1959. Asiatic clams as potential pests in California water supplies. Journal of the American Water Works Association 51:363–370.
- Joshi, R. C., R. H. Cowie, and L. S. Sebastian, editors. 2017. Biology and Management of Invasive Apple Snails. Philippine Rice Research Institute (PhilRice), Maligaya, Science City of Muñoz, Nueva Ecija, Philippines.
- Lodge, D. M., S. Williams, H. J. MacIsaac, K. R. Hayes, B. Leung, S. Reichard, R. N. Mack, P. B. Moyle, M. Smith, D. A. Andow, J. T. Carlton, and A. McMichael. 2006. Biological invasions: Recommendations for U.S. policy and management. Ecological Applications 16:2035–2054.
- Lowe, S., M. Browne, S. Boudjelas, and M. De Poorter. 2000. 100 of the world’s worst invasive alien species: A selection from the global invasive species database. Invasive Species Specialist Group, Aukland, New Zealand.
- Lv, S., Y. Zhang, H. X. Liu, L. Hu, K. Yang, P. Steinmann, Z. Chen, L. Y. Wang, J. Utzinger, and X. N. Zhou. 2009. Invasive snails and an emerging infectious disease: Results from the first national survey on Angiostrongylus cantonensis in China. PLoS Neglected Tropical Diseases 3:e368.
- McNeely, J. A. 2006. As the world gets smaller, the chances of invasion grow. Euphytica 14:5–15.
- Meyerson, L. A., and H. A. Mooney. 2007. Invasive alien species in an era of globalization. Frontiers in Ecology and the Environment 5:199–208.
- Pomacea snails (Mollusca: Pilidae) from Argentina to Asia. Micronesia 3:51–62.
- Moyle, P. B. 1999. Effects of invading species on freshwater and estuarine ecosystems. Pages 177–191 in O. T. Sandlund, P. J. Schei, and A. Viken, editors. Invasive Species and Biodiversity Management. Kluwer Academic Press, Amsterdam, Netherlands.
- Naylor, R. 1996. Invasions in agriculture: Assessing the cost of the golden apple snail in Asia. Ambio 25:443–448.
- Oya, S., Y. Hirai, and Y. Miyahara. 1987. Overwintering of the apple snail, Pomacea canaliculata Lamarck, in North Kyushu. Japanese Journal of Applied Entomology and Zoology 31:206–212.
- Perrings, C., K. Dehnen-Schmutz, J. Touza, and M. Williamson. 2005. How to manage biological invasions under globalization. Trends in Ecology and Evolution 20:212–215.
- Rawlings, T. A., K. A. Hayes, R. H. Cowie, and T. M. Collins. 2007.The identity, distribution, and impacts of non-native apple snails in the continental United States. BMC Evolutionary Biology 7:97–111.
- Schroeder, R. A., W. H. Orem, and Y. K. Kharaka. 2002. Chemical evolution of the Salton Sea, California: Nutrient and selenium dynamics. Hydrobiologia 473:23–45.
- Schoenherr, A. A. 2007. A Natural History of California. 2nd Edition. University of California Press, Berkeley, CA, USA.
- Sutherland, W. J., M. J. Bailey, I. P. Bainbridge, T. Brereton, J. T. A. Dick, J. Drewitt, N. K. Dulvy, N. R. Dusic, R. P. Freckleton, K. J. Gaston, P. M. Gilder, R. E. Green, A. L. Heathwaite, S. M. Johnson, D. W. Macdonald, R. Mitchell, D. Osborn, R. P. Owen, J. Pretty, S. V. Prior, H. Prosser, A. S. Pullin, P. Rose, A. Stott, T. Tew, C. D. Thomas, D. B. A. Thompson, J. A. Vickery, M. Walker, C. Walmsley, S. Warrington, A. R. Watkinson, R. J. Williams, R. Woodroffe, and H. J. Woodroof. 2008. Future novel threats and opportunities facing UK biodiversity identified by horizon scanning. Journal of Applied Ecology 45:821–833.
- Tseng, Y. T., H. C. Tsai, C. L. Sy, S. J. S. Lee, Y. H. Wann, Y. H. Wang, J. K. Chen, K. S. Wu, and Y. S. Chen. 2011. Clinical manifestations of eosinophilic meningitis caused by Angiostrongylus cantonensis: 18 years’ experience in a medical center in southern Taiwan. Journal of Microbiology, Immunology, and Infection 44:382–389.
- Wada, T., and K. Matsukura. 2009. Response to abiotic stress in Pomacea canaliculata with emphasis on cold tolerance. Pages 119–132 in R. C. Joshi and L. S. Sebastian, editors. Global Advances in Ecology and Management of Golden Apple Snails. Philippine Rice Research Institute, Nueva Ecija, Philippines.
- Williamson, M. H. 1996. Biological Invasions. Chapman and Hall, London, UK.
- Wood, T., P. Anurakpongsatorn, R. Chaichana, J. Mahujchariyawong, and T. Satapanajaru. 2005. Predation on freshwater bryozoans by the apple snail, Pomacea canaliculata, Ampularidae, an invasive species in Southeast Asia: A summary report. Denisia 16:283–286.
- Yeo, D. C. J. 2004. A new species of Esanthelphusa (Crustacea: Brachyura: Parathelphusidae) from Laos, and a redescription of Potam (Parathelphusa) dugasti Rathbun, 1902. The Raffles Bulletin of Zoology 52:219–226.

