RESEARCH NOTE
Jeff A. Alvarez1* and Jakob Woodall2
1 The Wildlife Project, P.O. Box 188888, Sacramento, CA 95818, USA![]() https://orcid.org/0000-0003-1875-6327
https://orcid.org/0000-0003-1875-6327
2 Stillwater Sciences, 2855 Telegraph Avenue, Berkeley, CA 94705, USA
![]() https://orcid.org/0009-0001-7369-6737
https://orcid.org/0009-0001-7369-6737
*Corresponding Author: jeff@thewildlifeproject.com
Published 27 December 2024 • doi.org/10.51492/cfwj.110.18
Key words: above ground, foraging, management implications, phenology, Spea hammondii, surveys, timing, western spadefoot
| Citation: Alvarez, J. A., and J. Woodall. 2024. Above ground phenology of terrestrial forms of western spadefoot with management recommendations. California Fish and Wildlife Journal 110:e18. |
| Editor: Jennifer Olson, Watershed Restoration Grants Branch |
| Submitted: 27 March 2024; Accepted: 10 October 2024 |
| Copyright: ©2024, Alvarez and Woodall. This is an open access article and is considered public domain. Users have the right to read, download, copy, distribute, print, search, or link to the full texts of articles in this journal, crawl them for indexing, pass them as data to software, or use them for any other lawful purpose, provided the authors and the California Department of Fish and Wildlife are acknowledged. |
| Competing Interests: The authors have not declared any competing interests. |
Species of the genus Spea, amphibians known as spadefoots, are secretive (Bragg 1965), generally remaining underground during dry periods and emerging during wet periods when conditions are more favorable (Storer 1925; Stebbins 1951, 1954; Stebbins and McGinnis 2012). Spea along the Pacific coast have been reported to generally spend the non-breeding season underground and emerge following fall and winter rain events to seek out vernal pools and similar habitat for reproduction (Storer 1925; Stebbins 1951, 1954; Thomson et al. 2015). After periods of rainfall, males migrate to breeding pools and vocalize to attract females (Stebbins 1954). Individual females arrive after males and likely breed only once a season, retreating to upland areas shortly after reproduction (Storer 1925). Upon the onset of dry, hot conditions, western spadefoots retreat underground for extended periods, emerging infrequently (Stebbins and McGinnis 2012; Flaxington 2021; Halstead et al. 2021). Because Spea are not known to be above ground for extended periods, they remain enigmatic for many aspects of their natural history (Bragg 1965; Jennings and Hayes 1994; Baumberger et al. 2019; Halstead et al. 2021).
The western spadefoot (Spea hammondii) is a small-bodied, fossorial, biphasic anuran native to California and Baja California in Mexico (Storer 1925; Stebbins and McGinnis 2012; Flaxington 2021). It has been listed as a Species of Special Concern by the state of California for three decades (Jennings and Hayes 1994; Thomson et al. 2015) and has been proposed for listing as threatened under the federal Endangered Species Act (USFWS 2023). The decline of this species in California has been attributed to habitat loss due to agriculture, largely in the Sacramento and San Joaquin Valley of California, and urban development in coastal southern California (Jennings and Hayes 1994; Thomson et al. 2015).
Since its initial description by Baird (1859), the majority of published work focused on the reproductive, developmental, or larval behavior (Brown 1976; Denver et. al. 1998; Morey and Reznick 2001; Morey and Reznick 2004; Ervin et al. 2005; Ervin and Cass 2007; Groff et al. 2012; Goldberg 2023). Significant portions of the natural history of the species were inferred from other, now distinct species, from which it has been separated (Neal et al. 2018; see also: Alvarez et al. 2024). Select aspects of the movement of western spadefoot in California have been reported, with the focus of these studies being the movement of adults from breeding sites to their self-excavated burrows (Baumberger et al. 2019; Halstead et al. 2021). Morey (1998) reported on the paucity of upland associated studies and suggested that there are conservation concerns related to an absence of understanding this aspect of the species life history. Baumberger et al. (2019) focused on western spadefoot home range and movement ecology in southern California and included information on movement triggers and burrow characteristics. They also reported that western spadefoot can be active above ground in the absence of rain events. These details greatly added to the knowledge of natural history of the species. Recently, Flaxington (2021) reported that western spadefoot may be active during humid periods from November to May but gave no clear details.
Through the use of online databases, we noted that western spadefoot has been anecdotally reported to be above ground more than previously known. Here, we add to the greater understanding of the natural history of western spadefoot by assembling and analyzing observations of this declining species in an attempt to establish its annual above-ground activity period.
We queried publicly available internet-based databases, including from published accounts, from authors’ observations, from Flickr, VertNet, CNDDB, H.E.R.P, iNaturalist, as well as smaller (i.e., no internet-based inventory) museum records, all known published accounts, anecdotal observations (via personal communications) from demonstrably knowledgeable individuals, and our own field observations. Each record collected, analyzed, and used in this study included the reported location and date of each observation. If the species, life stage, or date was ambiguous in any manner, the record was discarded. Any duplicated observations (i.e., the same individual recorded by multiple people on iNaturalist, or the same record on multiple platforms) were also discarded. We restricted our analysis to adult western spadefoot and adult forms (i.e., post-metamorphic and subadults).
We used the term “period” here and acknowledge that it is a time scale that is not clearly defined for the species. It can be in the range of months, with inter-annual variability. Herein, we use it to encompass a time-constrained behavior. For example, the estivation period has historically been considered nine to ten months (Stebbins and McGinnis 2012), but we content that may be shorter, a period of three months or less.
We acknowledge there are potential biases with using aggregate data in our analysis, including misidentification of individuals by the observer, inaccurate or imprecise location or temporal information, and the inherent sampling biases of random sightings. Data from iNaturalist was especially vulnerable to these shortcomings due to the high variability of observer skill. As such, each observation was verified by the researchers, whenever possible, before being included in this data set. Notwithstanding these potential biases, we believe our methodology supports our conclusions.
The data (n = 1041 records) we reviewed are broken down to the following sources: 1.1% (n = 12) from published accounts; 4.4% (n = 45) from authors’ observations; 4.4% (n = 46) from Flickr; 16.3% (n = 170) were attributed to VertNet; 17.3% (n = 180) from CNDDB; 21.1% (n = 220) from H.E.R.P; 27.3% (n = 285) from iNaturalist; and 7.9% (n = 83) from personal communications between the authors’ and individuals that work with the species. Some observations were clearly reported in two or more data sets. Those observations were filtered out of our data set as needed and attributed to the first reporting database in which we found the observation.
The records we evaluated span May 1908 to March 2024. The dataset suggested a peak in observations that occurs in March (19.80%, n = 207), with the majority of the observations reported February–May (66.3%, n = 690). This was followed by a decrease in reported observations from July through November (Fig. 1); characterized as the “less active period”—coinciding with what has generally been characterized as the non-breeding season (Storer 1925; Stebbins 1951, 1954; Stebbins and McGinnis 2012). The peak in observations from this study coincides with the peak reproductive period for western spadefoot, however, larvae have been reported during 11 of 12 months (≠ July) (Brown 1976; Ervin et al. 2005; Ervin and Cass 2007; Goldberg 2023), which makes the breeding season difficult to characterize.
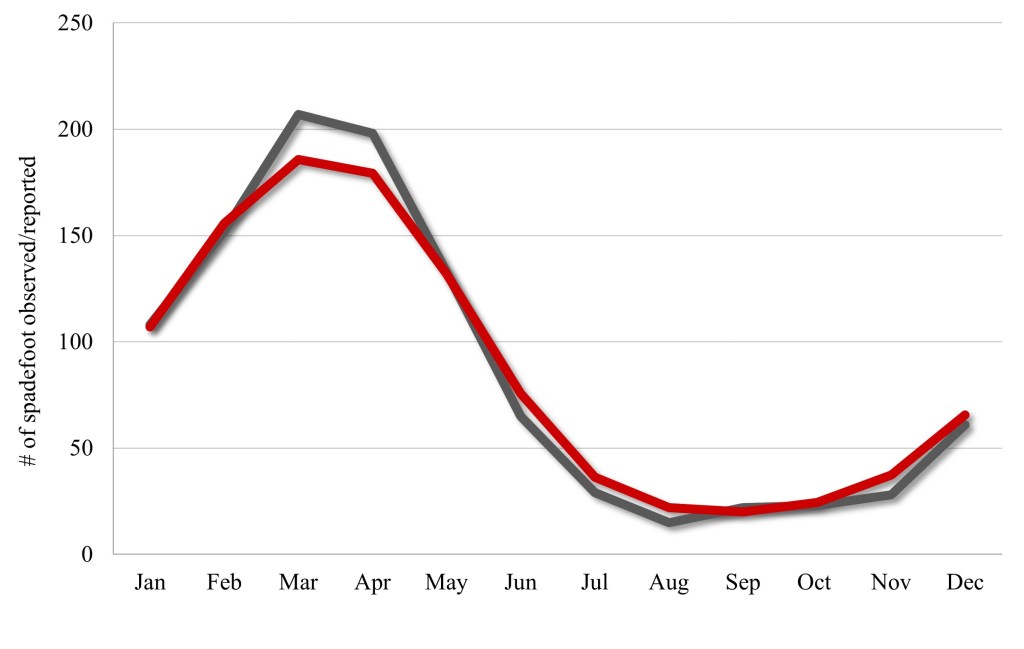
Nevertheless, it is the less active period that is critical for this species. We found that 34.7% of the reported observations (n = 345) were above ground during the less active period. This above ground activity appears to be focused on foraging and dispersal (pers. obs.). For example: we found 33 adults moving through grasslands, following sunset in April (post-breeding in this area), in Madera County, California, 26 days after the most recent rain event. These adults were as far as 100 m from the nearest breeding pond. During the same survey, we found 184 post-metamorphic western spadefoots dispersing from natal ponds, through open grassland areas.
Numerous authors have reported western spadefoot being essentially sessile, or nearly so, while underground (Storer 1925; Stebbins 1954; Morey 2005; Stebbins and McGinnis 2012; Groff et al. 2012). In contrast, Bragg (1965) reported that two species, the eastern spadefoot (Scaphiopus holbrookii) and Hurter’s spadefoot (S. hurteri) commonly returned to the surface, day after day, in order to feed, through much of the less active period. However, he conceded that, “habits of even closely related species may differ in different habitats”. We note that the western spadefoot occupies a semi-arid climate, while the species observed by Bragg (1965) occupy temperate climates. Storer (1925) attempted to test the response of western spadefoot to light in the lab—to determine if it was nocturnal. He reported that individual emerged from burrows on a nightly basis for the month that they were housed in captivity.
We note that there has been a putative acceptance that western spadefoot stays underground for long periods, yet this has not been demonstrated specifically for western spadefoot. We contend, however, that it is counterintuitive to believe that they could spend as much as nine or ten months with no above ground activity, as reported by Stebbins and McGinnis (2012), who suggest the species remains underground without emergence. It was not until recently that researchers suggested that the western spadefoot might have an “active period” where they may emerge periodically (Baumberger et al. 2019; Flaxington 2021; Halstead et al. 2021). We found that more than a third of observations reported were outside of the active period reported by Flaxington (2021), and further, our analysis demonstrates that there are records that support surface activity throughout the year for western spadefoot, which appears to be active above ground throughout the year, albeit likely intermittently and with specific peaks in above ground activity. Although more study is required to fully understand their emergence triggers, based on our own observations, we suggest that western spadefoot likely occupy shallow burrows during much of the year, emerging as needed, to forage, when local conditions support above ground activity. For example, over several seasons, we found numerous western spadefoots in Baja California, Mexico, in the month of May, presumably foraging during nighttime forays. These animals appeared above ground with no detectable increase in humidity, and no rainfall for at least 30 days prior to the observation (Fig. 2). They appeared to be buried in very shallow burrows, just beneath the surface. We believe these animals may emerge daily, or nearly so, during extended periods, as do other spadefoot species of the genus Scaphiopus (Bragg 1965).

This extensive above ground activity has important conservation implications in the range of western spadefoot. We believe that this speciesmay be susceptible to injury or mortality during any kind of surface activity that occurs during periods of above ground activity (i.e., elevated humidity, nighttime active periods, or other times following emergence). We speculate that sites where construction and ground disturbance occur during the day may cause surviving individuals to move during the night to relocate into suitable areas or occasionally unsuitable areas. This may cause potential entrapment conditions in trenches or excavations, or they may seek shelter under anthropogenic cover objects. This was noted when a western spadefoot was found in a construction site in Sacramento County, seeking refuge under a pile of riprap when the habitat in which it occupied was graded during the day. When the riprap was disturbed, the spadefoot was discovered (S. Foster, Foster Wildlife Surveys, pers. comm.).
Western spadefoot is a declining species and susceptible to elevated population declines due to habitat disturbance and alteration (Thompson et al. 2015; USFWS 2023). It is particularly vulnerable to injury or mortality during ground disturbance in areas where it occupies shallow, self-excavated burrows, due to the cryptic nature of this behavior (no observable ground-level sign of burrows). Baumberger et al. (2019) suggested an association between small mammal burrows and western spadefoot, but did not believe that this species used the burrows of California ground squirrels (Otospermophilus beecheyi) or Botta’s pocket gophers (Thomomys bottae), with which they were associated. However, Stebbins (1954) reported the use of kangaroo rat (Dipodomys sp.) burrows by western spadefoot. Further, Alvarez et al. (2023) reported spadefoot using the burrows of California ground squirrels, and we have made additional observations that suggest that this may be common. We noted that searches of California ground squirrel burrow systems during evening surveys, in occupied grasslands, commonly resulted in finding western spadefoot either on the ejecta piles or in the burrow entrances, in San Joaquin, Madera, and Los Angeles counties, California (pers. obs.). We believe that these burrow systems may provide open areas on which to forage, refuge sites in burrows, microsites that are easier to excavate for spadefoot individuals, and possibly sources of invertebrates for spadefoot (Alvarez 2023). Significant study needs to focus on the triggers that cause emergence, seasonal changes in depth of burrows, use of rodent burrows, and other aspects of this species natural history in order to assess its vulnerability to habitat disturbance. Until additional, detailed and thorough information has been reported that conclusively suggests timing and use of above ground habitats, places that support favorable ecological conditions should be considered occupied.
Although not studied here for western spadefoot (i.e., no clear trigger), we demonstrate that there is a decline in activity during late summer and fall where western spadefoot appears less frequently at the surface (Fig. 1). This pattern was also noted by Bragg (1965), who suggested that other species of spadefoot (i.e., Scaphiopus) have an emergence time during evening that becomes progressively later through the summer, until they no longer emerge in late summer and fall. This makes detection and conservation of this declining species a challenge as an absence of surface activity may be perceived as an absence of the species by unqualified surveyors.
Surveys for western spadefoot are typically associated with breeding periods, around breeding habitat, and on the larval life stage (pers. obs.). In addition to breeding period surveys, we would recommend spring and early summer nighttime upland survey efforts to assist with determining presence of the species. Evening upland surveys should avoid late summer and early fall time frames. When conducted in late spring and early summer, upland surveys should start approximately one hour after sunset (with later start times during the latter part of this time frame), and walking transects should include investigating burrow complexes of rodents and other species. We feel that it is critical to understand upland habitat use for this species in order to assess the value and extent of upland use for western spadefoot, not only for estivation and dispersal, but for frequent foraging bouts that can likely occur any month of the year.
Acknowledgements
We are grateful to the biologists that have shared their observations of Spea hammondii during constructive conversation about the species. We thank S. M. Foster of Foster Wildlife Surveys, N. Kleponis of California State Parks, M. Olson of Westervelt Ecological Services, A. Peralta García and J. H. Valdez-Villavicencio of Conservación de Fauna del Noroeste, Ensenada, Baja California for sharing their observations. We also thank G. Padgett-Flohr who facilitated access to the Tesoro Highlands site in Los Angels County, CA, and to J. Howe, of Westervelt Ecological Services, who facilitated access to the HOG Hills Preserve, Madera County, CA.
Literature Cited
- Alvarez, J. A. 2023. Role of burrow systems of California ground squirrel burrows (Otopspermophilus beecheyi) in sustaining native wildlife. Western Wildlife 10:47–55.
- Alvarez, J. A., and T. S. Kerss. 2023. Microhabitat use by post-metamorphic western spadefoot (Spea hammondii) in central California. Sonoran Herpetologist 36:13–14.
- Alvarez, J. A., A. Peralta-García, and J. H. Valdez Villavicencio. 2024. Putative cannibalism in the western spadefoot (Spea hammondii) in northern Baja California, México. Sonoran Herpetologist 37:34–36.
- Baird, S. F. 1859. Report upon the reptiles of the route. Reports of Explorations and Surveys, to Ascertain the Most Practicable and Economical Route for a Railroad from the Mississippi River to the Pacific Ocean. Made under the direction of the Secretary of War in 1856–6. Volume 10, Williamson’s Route. Zoological Report, Part 6, Number 4. A. O. P. Nicholson, Washington, D.C., USA.
- Baumberger, K. L., M. V. Eitzel, M. E. Kirby, and M. H. Horn. 2019. Movement and habitat selection of the western spadefoot (Spea hammondii) in southern California. PLOS ONE 14:e0222532.
- Bragg, A. N. 1965. The spadefoot toads in Oklahoma with our summary of our knowledge of the group: II. American Naturalist 79:52–72.
- Brown, H. A. 1976. The status of California and Arizona populations of the western spadefoot toads (genus Scaphiopus). Contributions in Science, Los Angeles County Museum of Natural History 286:1–15.
- Denver, R. J., N. Mirhadi, and M. Phillips. 1998. Adaptive plasticity in amphibian metamorphosis: response of Scaphiopus [Spea] hammondii tadpoles to habitat desiccation. Ecology 79:1859–1872.
- Ervin, E. L., C. D. Smith, and S. V. Christopher. 2005. Spea hammondii (western spadefoot): reproduction. Herpetological Review 36:309–310.
- Ervin, E., and T. Cass. 2007. Spea hammondii (western spadefoot): reproductive pattern. Herpetological Review 38:196–197.
- Flaxington, W. C. 2021. Amphibians and Reptiles of California: Field Observations, Distributions, and Natural History. Fieldnotes Press, Anaheim, CA, USA.
- Goldberg, S. R. 2023. Notes on reproduction of western spadefoot toads, Spea hammondii (Anura: Scaphiopodidae). Sonoran Herpetologist 36:10–12.
- Groff, L. A., W. G. Duffy, S. N. Kahara, and S. J. Chapin. 2012. Temporally irregular breeding of western spadefoot toads (Spea hammondii) in managed wetlands. Northwestern Naturalist 93:79–83.
- Halstead, B. J., K. L. Baumberger, A. R. Backlin, P. M. Kleeman, M. N. Wong, E. A. Gallegos, J. P. Rose, and R. N. Fisher. 2021. Conservation implications of spatiotemporal variation in the terrestrial ecology of western spadefoots. Journal of Wildlife Management 85:1377–1393.
- Jennings, M. R., and M. P. Hayes. 1994. Amphibian and Reptile Species of Special Concern in California. California Department of Fish and Game, Fisheries Division. Rancho Cordova, CA, USA.
- Morey, S. R. 2005. Spea hammondii (Baird, 1859, “1857”) Western Spadefoot. Pages 514-517 in M. Lannoo, editor. Amphibian Declines; The Conservation Status of United States Species, University of California Press, Berkeley, CA, USA.
- Morey, S. R. 1998. Pool duration influences age and body mass at metamorphosis in the western spadefoot toad: implications for vernal pool conservation. Pages 86–91 in C. W. Witham, E. T. Bauder, D. Belk, W. R. Ferren, Jr., and R. Ornduff, editors. 1996. Ecology, Conservation, and Management of Vernal Pool Ecosystems – Proceedings from a 1996 Conference. California Native Plant Society, Sacramento, CA, USA.
- Morey, S., and D. Reznick. 2001. Effects of larval density on post-metamorphic spadefoot toads (Spea hammondii). Ecology 82:510–522.
- Morey, S. R., and D. N. Reznick. 2004. The relationship between habitat permanence and larval development in California spadefoot toads: field and laboratory comparisons of developmental plasticity. Oikos 104:172–190.
- Neal, K. M., B. B. Johnson, and H. B. Shaffer. 2018. Genetic structure and environmental niche modeling confirm two evolutionary and conservation units within the western spadefoot (Spea hammondii). Conservation Genetics 19:937–946.
- Stebbins, R. C. 1951. Amphibians of Western North America. McGraw-Hill Publishing Company, New York, NY, USA.
- Stebbins, R. C. 1954. Amphibians and Reptiles of Western North America. McGraw-Hill Publishing Company, New York, NY, USA.
- Stebbins, R. C., and S. M. McGinnis. 2012. Field Guide to Amphibians and Reptiles of California, revised edition. University of California Press, Berkeley, CA, USA.
- Storer, T. I. 1925. A synopsis of the Amphibia of California. University of California Publications in Zoology 27:1–342.
- Thomson, R. C., A. N. Wright, and H. B. Shaffer. 2015. California Amphibian and Reptile Species of Special Concern. University of California Press, Berkeley, CA, USA.
- U.S. Fish and Wildlife Service (USFWS). 2023. Endangered and Threatened Wildlife and Plants: Threatened Status With Section 4(d) Rule for the Northern and Southern Distinct Population Segments of the Western Spadefoot. Federal Register 88:84252–84278.

