FULL RESEARCH ARTICLE
John D. Wehausen1*, Christopher J. O’Brien2, and Dale R. McCullough3
1 University of California, White Mountain Research Center, 3000 East Line Street, Bishop, CA 93514, USA![]() https://orcid.org/0000-0002-2926-9682
https://orcid.org/0000-0002-2926-9682
2 Lassen Community College, 478-200 Highway 139, Susanville, CA 96130, USA
3 University of California, Berkeley, Department of Environmental Science, Policy, and Management, Berkeley, CA 94720, USA![]() https://orcid.org/0000-0001-8072-4188
https://orcid.org/0000-0001-8072-4188
*Corresponding Author: jdwehausen@gmail.com
Published 27 December 2024 • doi.org/10.51492/cfwj.110.19
Abstract
Tooth cementum rings often have been considered the most reliable method for aging larger mammals. Tests of this approach have used samples of known ages, but to ensure objectivity this needs to be carried out as truly blind tests. That has not always been the case, including for North American wild sheep in studies that concluded that cementum ring aging provided highly accurate ages. This study re-examined cementum ring aging accuracy for bighorn sheep (Ovis canadensis) via a blind test of known-age samples from two ecologically different regions in California, using three different laboratory techniques (histological, petrographic, and grinding), and different individuals applying each of those methods. Overall, there were considerably more errors than previously reported. The grinding method performed particularly poorly, but statistical analyses of aging errors for the other two methods found differences between those methods and between the two data sets used. While the histological method had the highest overall rate of correct ages at 31%, its error range was twice that of the petrographic method. Given the error rates we found, cementum-based ages of bighorn sheep should not be treated as accurate ages. It is therefore unlikely that cementum-based ages will improve ages based on horn rings for most male specimens. Cementum-based ages may be useful for female bighorn sheep older than 4-6 years because of difficulties in aging by horn rings.
Key words: bighorn sheep, Ovis canadensis, cementum ring ages
| Citation: Wehausen, J. D., C. J. O’Brien, and D. R. McCullough. 2024. Reliability of tooth cementum rings to age bighorn sheep: a blind test. California Fish and Wildlife Journal 110:e19. |
| Editor: Thomas Connor, Wildlife Branch |
| Submitted: 21 February 2024; Accepted: 28 May 2024 |
| Copyright: ©2024, Wehausen et al. This is an open access article and is considered public domain. Users have the right to read, download, copy, distribute, print, search, or link to the full texts of articles in this journal, crawl them for indexing, pass them as data to software, or use them for any other lawful purpose, provided the authors and the California Department of Fish and Wildlife are acknowledged. |
| Competing Interests: The authors have not declared any competing interests. |
Introduction
Age has long been recognized as an important variable in studies of animal population ecology, and numerous methods have been developed to estimate age, depending on the species (Taber 1971). For many mammals, tooth cementum rings have been thought to provide the most reliable ages (Thomas 1977). Horn rings for caprinae species frequently offer an alternative source of ages. For bighorn sheep (Ovis canadensis), horn rings of males in northern climates provide an accurate measure of age (Geist 1966). However, this method is considerably less reliable for females that grow small horns often with unclear rings later in life. Consequently, tooth cementum ring aging offers an attractive alternative for many bighorn sheep.
Gonzales (1976) reported on samples from ten desert bighorn sheep males from Baja California, comparing ages from horn rings and tooth cementum rings. That study found a close correspondence between the ages from the two data sources, but what teeth were used was not stated, and there was no indication of independence between the two data sources. Turner (1977) analyzed first incisors from 202 North American wild sheep representing a wide variety of habitats, including two species and both sexes. Known ages were determined from horn annuli for most samples by multiple people providing samples, but also by known birth dates for 12 bighorn sheep. That study reported a high accuracy for cementum ring ages in which error up to age seven almost never exceeded 0.5 years. For older ages the error level increased, but only 9% of 80 samples were incorrect by >1 year, and the worst specimens deviated from the correct age by only slightly more than one year. While that author stated that all ages from teeth were determined prior to comparison with ages from other sources, the analysis was not blind in that the prior assigned ages were available to the investigator generating the tooth ages, and that person compiled and analyzed the results.
The first author had reason to question the reliability of tooth cementum aging in bighorn sheep after receiving an estimated age from a commercial lab specializing in such aging for a tooth from a desert bighorn (O. c. nelsoni) ewe from California that considerably exceeded maximum longevities documented for any North American wild sheep. That concern led to this study in which we report on a completely blind test of cementum ring aging of bighorn sheep using three different laboratory techniques and two data sets from different ecosystems in California.
Methods
Study Area and Specimens Used
We used two sample sets of bighorn sheep of known age from different regions of eastern California. A southern sample set came from desert bighorn sheep in three nearby mountain ranges of the Eastern Mojave Desert about 100 km east of Barstow: the Marble Mountains, Granite Mountains, and Old Dad Mountain (Fig. 1). Highest elevations in those mountain ranges are 1171–2054 m. That sample consisted of skulls from seven females first caught at ages <4 years when their ages were determined definitively through tooth replacement (Deming 1952; Taber 1971) and horn rings. Those individuals carried functional telemetry collars until their deaths; thus, their ages at death were known with certainty. The second, northern, sample set consisted of nine male skulls that were chosen because horn annuli were particularly distinct, making ages at death also unambiguous. Seven of these were from Sierra Nevada bighorn sheep (O. c. sierra) and two were from desert bighorn sheep, one from the White Mountains, and one from Dry Mountain (Fig. 1). In contrast with the bighorn sheep sampled in the eastern Mojave Desert, those sampled from the Sierra Nevada and White Mountains maximize annual nutrient intake by migrating seasonally between low elevation winter ranges at the eastern base of the Sierra Nevada and the western base of the White Mountains and extensive alpine habitats above 3450 m elevation (Wehausen and Hansen 1988). The maximum elevation of Dry Mountain is well below that alpine zone at 2644 m. For each set of samples, ages at death ranged from 3 to 12 years (3, 4, 6, 8, 9, 11, 12 for the southern data set, and 3, 5, 6, 7, 8, 8, 9, 11, 12 for the northern data). Teeth were carefully removed from skulls and cleaned so as not to damage cementum.

Data Development
For wild ungulates, cementum is deposited on teeth during forage growing seasons when nutrient intake is high but is punctuated by a potentially long period of the year when diet quality is low. That period of low diet quality produces narrow dark bands in the cementum that allow age determination (Hemming 1969). Fancy (1980) identified three basic laboratory methods to detect that banding pattern, referred to here as histological, petrographic, and grinding. For earlier studies of bighorn sheep, Gonzales (1976) used a method similar to the petrographic method, while Turner (1977) used the histological method. We used all three methods in this study. In the histological method, teeth are decalcified, rinsed, dehydrated, cleared, embedded and sectioned with a microtome into numerous slices, mounted on slides, stained, sealed with resin under a coverslip, and inspected microscopically under transmitted light (Hamlin et al. 2000). In the petrographic method, sequentially cut and polished surfaces of a tooth embedded in epoxy are attached to microscope slides, re-sectioned, then polished with silicon carbide grit until desired structures are evident under cross polarized light (Morris 1972). In the grinding method, molars are cut in half with a diamond saw, then repeatedly ground and inspected with reflected light until the optimal cementum history is evident. Alcohol on the ground surface can improve visibility of the cementum pattern. In the histological and petrographic methods, the entire roots of teeth are available for examination. In the grinding method, only the region between roots beneath the crown referred to as the “cementum pad” (Sargeant and Pimlott 1959; Wolfe 1969; Spinage 1973), “root saddle” (Welsh 1975), or “interradicular arch” (Spinage 1976) is examined.
In this study, the histological, petrographic, and grinding methods were carried out, respectively, by Matson’s Lab (www.MatsonsLab.com), the second author, and the third author, all of which had considerable experience using their respective methods for other species. The first author assembled the teeth and was the only one who knew the assigned ages; Matson’s Lab was not informed that the teeth provided were part of a test. Different teeth were used depending on methodological preferences, subject to availability. Matson’s lab received 11 first incisors (I1), 4 lower first premolars (PM1), and 1 lower first molar (M1). Only molars were used for the other two methods; the first molars were used except for one second molar (M2) for the petrographic method and two second molars for the grinding method. Upper molars were used for these latter two methods because of a potentially better cementum record than in lower molars (McCullough and Beier 1986). For each tooth, all investigators followed the approach of Matson’s Lab of providing the number of cementum rings and a range of rings considered to include the correct number where uncertainty existed.
Cementum ring counts were adjusted for estimates of ages at death on the basis of the tooth used relative to tooth replacement patterns (Deming 1952; Taber 1971) and known seasons of deaths relative to seasons when annuli are laid down. To obtain age estimates, the following integers were added to the number of rings as a function of the tooth examined: I1, 1; PM1, 2; M1, 0; M2, 1.
We analyzed results from the two sample sets both separately and combined. Analyses also were made separately for the point estimates and estimate ranges. Calculated for each data set were: (1) percentage of samples giving the correct age; (2) range of deviations from correct ages measured in years; (3) mean deviation of absolute values (sign ignored); and (4) bias, calculated as mean deviation with sign included. Deviations from correct values were calculated as estimated minus known ages; thus, overestimates produced positive values and underestimates negative values in years. Estimated age ranges that included the known age were classified as correct, thus zero deviation. For samples with ranges that did not include the known age, the deviation was the number of years between the known age and the closest range value. We analyzed deviation results in SYSTAT via repeated measures ANOVAs to investigate differences between the two data sets and laboratory methods, and paired t-tests for the combined data.
We investigated potential correlations between known ages and deviations of estimated ages and age ranges from known ages. This was done for the raw deviation data and for absolute values of deviations. Because point estimates and estimated ranges for two data sets were involved, we ran eight separate Pearson correlation tests in Excel for each lab method.
Results
For two of the seven southern samples, the grinding method failed to find any cementum rings and therefore failed to produce age estimates. Other methods located cementum rings for all teeth, as did the grinding method for the northern sample set. The grinding method produced no correct ages for the five southern samples with data, but one age range included the correct age. For the northern samples, the grinding method produced no correct ages for point estimates or for age ranges. The histological method correctly estimated ages of 29% of southern samples and 33% of northern samples, which increased respectively to 43% and 56% for age ranges. For the petrographic method, point estimates were correct for 14% of southern and 11% of northern samples, which respectively increased to 57% and 44% for age ranges (Table 1).
Table 1. Comparison of accuracy and precision of three methods of tooth cementum ring aging of bighorn sheep (Ovis canadensis) for two data sets from California: a southern set (S) of seven females from the Eastern Mojave Desert and a northern set (N) of nine males from the Sierra Nevada and nearby ranges to the east (Fig. 1). Methods were grinding (G), histological (H), and petrographic (P). Estimate types are exact age (point) and range of ages (range). Deviations are differences of estimates and estimate ranges from known ages in years with negative values representing underestimates. Mean absolute deviation is the mean of absolute values of deviations. Bias is the mean of deviations with sign included. For the southern samples, the grinding method produced no ages for two of the seven specimens and deviations and bias were based on the remaining five, whereas the % correct was based on all seven.
| Sample Set | Estimate Type | Method | % Correct | Deviation Range Low | Deviation Range High | Deviation Range Width | Mean Absolute Deviation | Bias |
| S | point | G | 0 | –6 | –1 | 5 | 2.8 | –2.8 |
| S | point | H | 29 | –4 | 0 | 4 | 2.0 | –2.0 |
| S | point | P | 14 | –2 | +2 | 4 | 1.3 | –0.1 |
| S | range | G | 14 | –5 | 0 | 5 | 2.2 | –2.2 |
| S | range | H | 43 | –4 | 0 | 4 | 1.6 | –1.6 |
| S | range | P | 57 | –2 | +1 | 3 | 0.7 | –0.4 |
| N | point | G | 0 | –2 | +11 | 13 | 3.3 | +2.4 |
| N | point | H | 33 | –2 | +4 | 6 | 1.2 | +0.1 |
| N | point | P | 11 | –1 | +2 | 3 | 0.9 | +1.1 |
| N | range | G | 0 | –2 | +8 | 10 | 2.7 | +1.8 |
| N | range | H | 56 | –2 | +2 | 4 | 0.7 | 0.0 |
| N | range | P | 44 | –1 | +2 | 3 | 0.4 | +0.7 |
| Both | point | G | 0 | –6 | +11 | 17 | 3.1 | +0.6 |
| Both | point | H | 31 | –4 | +4 | 8 | 1.6 | –0.8 |
| Both | point | P | 12 | –2 | +2 | 4 | 1.2 | +0.4 |
| Both | range | G | 6 | –5 | +8 | 13 | 2.5 | +0.4 |
| Both | range | H | 50 | –4 | +2 | 6 | 1.1 | –0.7 |
| Both | range | P | 31 | –2 | +2 | 4 | 0.7 | +0.1 |
Differences between point estimates and correct ages ranged from –6 to +11 for the grinding method, –4 to +4 for the histological method, and –2 to +2 for the petrographic method; for estimated age ranges these were, respectively, –5 to +8, -4 to +2, and -2 to +2 (Table 1). Average absolute value deviations of point age estimates from correct ages ranged from a low of 0.9 for the petrographic methods to a high of 3.3 for the grinding method (Table 1). For estimated age ranges, these average deviations ranged from 0.4 for the petrographic method to 2.7 for the grinding method (Table 1). For our 24 tests, we found no significant correlations between known ages and deviations of estimated ages and age ranges from known ages (P= 0.28–0.99).
Bias values indicated an overall tendency to underestimate the ages of the southern samples and to overestimate the ages of northern samples (Table 1; Fig. 2). Given a notable difference in results between the grinding and the other two methods, only the results from the histological and petrographic methods were further tested via ANOVA. First was a 2-way repeated measures ANOVA of deviations of point estimates from known ages that provided statistical support for differences between the two sample sets (F1,14 = 5.193, P = 0.039) and between the two methods (F1,14 = 10.511, P = 0.006), but no interaction effect (F1,14 = 1.764, P = 0.205). This is reflected in the bias results. For the southern data set, the petrographic method had almost no bias (–0.1), while the histological method had a bias of –2.0. In contrast, for the northern data set the histological method had almost no bias (+0.1) compared with +1.1 for the petrographic data (Table 1). For point estimates, the petrographic method exhibited a narrower range of deviations of estimated ages relative to known ages (–2 to +2 vs –4 to +4; Table 1; Fig. 1).
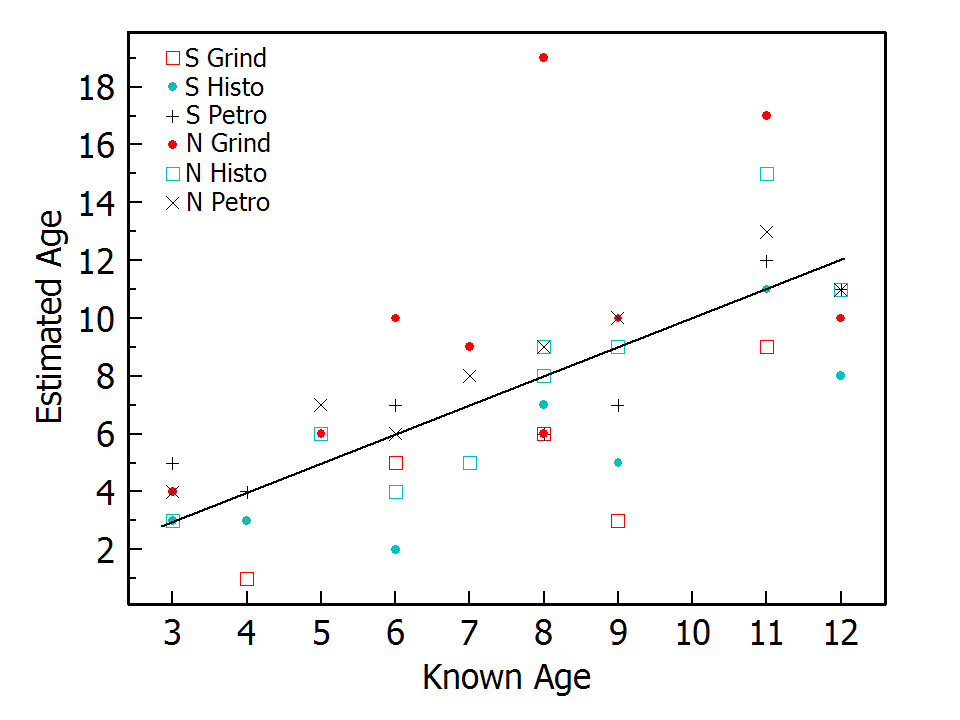
A 2-way repeated measures ANOVA of deviations of estimated age ranges from known ages again had no interaction effect (F1,28 = 0.993, P = 0.336) and again support for differences between sample sets (F1,14 = 5.859, P= 0.03), and between methods (F1,14 = 5.129, P= 0.04). This also reflects differences in bias results between methods and data sets (Table 1), and again the petrographic method exhibited a narrower range of deviations (–2 to +2) compared with the histological method (–4 to +2). These deviation range differences are reflected in higher means of absolute deviations for the histological method compared with the petrographic method (Table 1).
When the two sample sets were combined, point estimates for the histological method underestimated ages by 0.8 years on average, while the petrographic method overestimated by 0.4 years (Table 1; Fig. 2)—a difference supported by a paired t-test (t15 = –3.024.06, P = 0.009). For estimate ranges of the combined data sets, the histological method underestimated ages by 0.7 years on average, while the petrographic method overestimated by 0.1 years (Table 1)—a difference also with statistical support (t15 = –2.158, P = 0.048). For the combined data, the petrographic method again exhibited narrower deviation bands than the histological method for point and range data (Table 1).
Discussion
While none of the methods we tested performed well in terms of percentages of ages estimated correctly, the grinding method clearly performed worse than the other two methods. Reliance on the limited area between the roots of the teeth examined in the grinding method is a likely reason. While this method has provided reliable cementum patterns for aging white-tailed deer (McCullough 1979; McCullough and Beier 1986), the cementum in this area is cellular cementum that can provide a less reliable record of seasonal variation than acellular cementum found near the enamel-cementum junction (tooth cervix) on the side of the tooth. Cellular cementum in the root saddle develops rapidly in response to longitudinal displacement of the tooth, which occurs as a function of multiple factors over the life of an ungulate (Lieberman and Meadow 1992). This variation can lead to patterns that are difficult to interpret compared with acellular cementum. Ransom (1966) noted the need to look at the root of some older specimens to find a full cementum pattern, and many ungulate studies have used cementum patterns on other tooth surfaces (Low and Cowan 1963; Gilbert 1966; Keiss 1969; McCutchen 1969; Gasaway et al. 1978; Hamlin et al. 2000). The apparent lack of a good cementum record in the molar tooth saddle of deer on a high nutritional plane associated with low density (McCullough 1996) may not hold for acellular cementum at the tooth cervix.
For the two best methods in our results, we found the percentage of ages correctly estimated not to be the best criterion for evaluating laboratory methods. The histological method clearly outperformed the petrographic method in percent of correct point estimates for both data sets and for those sets combined (Table 1), but the petrographic method consistently provided age estimates with less overall error as measured by mean absolute deviation (Table 1; Fig. 2).
Early literature on cementum aging had a focus on methods and teeth used, typically using known-age samples in efforts to find the best method. While some early studies did this without reporting on accuracy of the method (Gilbert 1966; Lockard 1972), others (Low and Cowan 1963; Ransom 1966; McCutchen 1969; Keiss 1969) reported high accuracy of their methods, but with no evidence that tests were blind—a concern raised by Jacobson and Reiner (1989). Gasaway et al. (1978) appear to have used a blind approach (although not so stated), and others since then also have used that approach to provide more reliable assessments of accuracy (Moffitt 1998; Hamlin et al. 2000; Boertje et al. 2015). Our study adds to those efforts and provides a more realistic assessment of the potential error involved in ages of bighorn sheep estimated from tooth cementum rings relative to earlier publications (Gonzales 1976; Turner 1977). Hamlin et al. (2000) found a considerably higher accuracy of cementum aging for three North American cervid species than our results for bighorn sheep. Our results are more similar to Moffitt’s (1998) results for bison (Bison bison), another bovid species. It is not clear, however, that this accuracy difference is due to taxonomic family, given that Miura (1985) reported a high accuracy of cementum aging for a different bovid species, Japanese serow (Capricornis crispus).
The cause of formation of cementum rings has been debated, with environmental variables often cited as the cause for two annual (Spinage 1973, 1976, 1979), indistinct (Thomas and Bandy 1973), or missing (Klingel and Klingel 1966; Spinage 1971) rings. Others have demonstrated that methodological approaches and the variability in cementum tissues may be a greater influence (O’Brien 1994, 2001; Lieberman 1994). While our investigation has added information on methodological differences, we also found different results from different ecosystems. Given that we sampled only part of the variation in ecological settings inhabited by bighorn sheep, it is likely that there is yet more cementum ring variation in this species.
A conclusion from our findings is that cementum-based ages for bighorn sheep may not be appropriate for analyses requiring correct ages. Instead, it might be necessary to break ages into a set of age ranges for which some error is less important. Cementum-based ages will continue to provide important additional information for sex and age classes in which ages based on the combination of tooth replacement and horn rings can be unreliable. That is frequently the case for females older than 4–6 years old, perhaps especially in colder environments in which females grow less horn compared with most desert bighorn sheep. In contrast, male horns of North American wild sheep provide reliable ages in colder regions with distinct and consistent seasonality in horn growth. For such specimens, it is unlikely that cementum-based ages will provide useful additional information, as our results demonstrated.
In desert environments, temporally predictable spring nutrient pulses vary greatly in magnitude across years due to highly variable cool season rainfall, but there also can be a warm season nutrient pulse when sufficient summer rains occur (Wehausen 2005). This variation can influence horn growth, sometimes leading to two horn rings in a year, especially in the Sonoran Desert. In such environments male horn rings may be less reliable, but cementum-based ages also may suffer from the same sources of potential error and perhaps add no additional useful information.
Acknowledgments
Kathleen Anderson kindly created the map. We thank three reviewers for helpful comments.
Literature Cited
- Boertje, R. D., M. M. Ellis, and K. A. Kellie. 2015. Accuracy of moose age determinations from canine and incisor cementum annuli. Wildlife Society Bulletin 39:383–389.
- Deming, O. V. 1952. Tooth development of the Nelson bighorn sheep. California Fish and Game 38:523–529.
- Fancy, S. G. 1980. Preparation of mammalian teeth for age determination by cementum layers: a review. Wildlife Society Bulletin 8:242–248.
- Gasaway, W. C., D. B. Harkness, and R. A. Rausch. 1978. Accuracy of moose age determinations from incisor cementum layers. Journal of Wildlife Management 42:558–563.
- Geist, V. 1966. Validity of horn segment counts in aging bighorn sheep. Journal of Wildlife Management 30:634–635.
- Gilbert, F. F. 1966. Aging white-tailed deer by annuli in the cementum of the first incisor. Journal of Wildlife Management 30:200–202.
- Gonzales, M. 1976. Determination of the age of the bighorn sheep. Desert Bighorn Council Transactions 20:23–24.
- Hamlin, K. L., D. F. Pac, C. A. Sime, R. M. DeSimone, and G. L. Dusek. 2000. Evaluating the accuracy of ages obtained by two methods for Montana ungulates. Journal of Wildlife Management 64:441–449.
- Hemming, J. E. 1969. Cementum deposition, tooth succession, and horn development as criteria of age in Dall sheep. Journal of Wildlife Management 33:552–558.
- Jacobson, H. A., and R. J. Reiner. 1989. Estimating age of white-tailed deer: tooth wear versus cementum annuli. Proceedings of the Southeastern Fish and Wildlife Conference 43:286–291.
- Keiss, R. E. 1969. Comparison of eruption-wear patterns and cementum annuli as age criteria in elk. Journal of Wildlife Management 33:175–180.
- Klingel, H. and U. Klingel 1966. Tooth development and age determination in the plains zebra (Equus quagga boehmi Matschie). Der Zoologische Garten 33:34–54.
- Lieberman, D. E. 1994. The biological basis for seasonal increments in dental cementum and their application to archaeological research. Journal of Archaeological Science 261:1162–1164.
- Lieberman, D. E., and R. H. Meadow. 1992. The biology of cementum increments (with an archeological application). Mammal Review 22:57–77.
- Lockard, G. R. 1972. Further studies of dental annuli for aging white-tailed deer. Journal of Wildlife Management 36:46–55.
- Low, W. A. and I. McT. Cowan. 1963. Age determination of deer by annular structure of dental cementum. Journal of Wildlife Management 27:466–471.
- McCullough, D. R. 1979. The George Reserve Deer Herd. University of Michigan Press, Ann Arbor, MI, USA.
- McCullough, D. R. 1996. Failure of the tooth cementum aging technique with reduced population density of deer. Wildlife Society Bulletin 24:722–724.
- McCullough, D. R. and P. Beier. 1986. Upper vs. lower molars for cementum annuli age determination of deer. Journal of Wildlife Management 50:705–706.
- McCutchen, H. E. 1969. Age determination of pronghorns by the incisor cementum. Journal of Wildlife Management 33:172–175.
- Miura, S. 1985. Horn and cementum annulations as age criteria in Japanese serow. Journal of Wildlife Management 49:152–156.
- Moffitt, S. A. 1998. Aging bison by the incremental cementum growth layers in teeth. Journal of Wildlife Management 62:1276–1280.
- Morris, P. 1972. A review of mammalian age determination methods. Mammal Review 2:69–104.
- O’Brien, C. J. 1994. Determining seasonality and age in East African archaeological faunas: an ethnoarchaeological application of cementum increment analysis. Dissertation, University of Wisconsin, Madison, WI, USA.
- O’Brien, C. J. 2001. A re-evaluation of dental increment formation in east African mammals: implications for wildlife biology and zooarchaeology. ArchaeoZoologia 11:43–64.
- Ransom, A. B. 1966. Determining age of white-tailed deer from layers in cementum of molars. Journal of Wildlife Management 30:197–199.
- Sargeant, D. E. and D. H. Pimlott. 1959. Age determination in moose from sectioned incisor teeth. Journal of Wildlife Management 23:315–321.
- Spinage, C. A. 1971. Geratodontology and horn growth of the impala (Aepyceros melampus). Journal of Zoology, London 164:209–225.
- Spinage, C. A. 1973. A review of the age determination of mammals by means of teeth, with special reference to Africa. East African Wildlife Journal 11:165–187.
- Spinage, C. A. 1976. Incremental cementum lines in the teeth of tropical African mammals. Journal of Zoology, London 178:117–131.
- Spinage, C. A. 1979. A model for predicting age in ungulates. Pages 33–40 in S. S. Ajayi and L. B. Halstead, editors. Wildlife Management in Savanna Woodlands. Taylor and Francis LTD, London, UK.
- Taber, R. D. 1971. Criteria of sex and age. Pages 325–401 in R. H. Giles, editor. Wildlife Management Techniques. The Wildlife Society, Washington, D.C., USA.
- Thomas, D. C. 1977. Metachromatic staining of dental cementum for mammalian age determination. Journal of Wildlife Management 41:207–210.
- Thomas, D. C. and P. J. Bandy 1973. Age determination of wild black-tailed deer from dental annulations. Journal of Wildlife Management 37:232–235.
- Turner, J. C. 1977. Cementum annulations as an age criterion in North American sheep. Journal of Wildlife Management 41: 211–217.
- Wehausen, J. D. 2005. Nutrient predictability, birthing season, and lamb recruitment for desert bighorn sheep. Pages 37–50 in J. Goerrissen and J. M André, editors. Sweeney Granite Mountains Desert Research Center 1978–2003: A Quarter Century of Research and Teaching. University of California Natural Reserve Program, Riverside, CA, USA.
- Wehausen, J. D., and M. C. Hansen. 1988. Plant communities as the nutrient base of mountain sheep populations. Pages 256–268 in C. A. Hall, Jr., and V. Doyle-Jones, editors. Plant Biology of Eastern California. Natural History of the White-Inyo Range Symposium Volume 2. White Mountain Research Station, Bishop, CA, USA.
- Welsh, D. A. 1975. Population, behavioral and grazing ecology of the horses of Sable Island, Nova Scotia. Dissertation, Dalhousie University, Halifax, Novia Scotia, Canada.
- Wolfe, M. L. 1969. Age determination in moose from cementum layers of molar teeth. Journal of Wildlife Management 33:428–431.

