FULL RESEARCH ARTICLE
David M. Laabs1*, Mark L. Allaback1, and Donald R. Mitchell2
1 Biosearch Environmental Consulting, P.O. Box 1220, Santa Cruz, CA 95061, USA
2 ECORP Consulting, Inc., 215 N 5th St., Redlands, CA 92374, USA
*Corresponding Author: laabs@calcentral.com
Published 1 July 2022 • http://www.doi.org/10.51492/cfwj.108.10
Abstract
The San Joaquin pocket mouse (Perognathus inornatus) is endemic to California and currently includes three subspecies (P. i. inornatus, neglectus, and psammophilus). P. inornatus from the western Mojave Desert have been assigned to the subspecies neglectus based on similar morphology. During the course of live-trapping projects between 1990 and 2017, we captured P. inornatus at several locations in the Mojave Desert and gathered information on habitat associations, relative abundance, seasonal activity, and field identification. P. inornatus was found at elevations ranging from 668–1,109 m above mean sea level in creosote bush scrub, allscale scrub, Joshua tree woodland, rubber rabbitbrush scrub, spinescale scrub, and California juniper woodland. The capture locations and our review of museum specimens indicate that, at a minimum, the species’ range in the Mojave Desert encompasses approximately 7,000 km2 primarily in the Antelope Valley, extending at least as far north as the Rand Mountains, east to the vicinity of Hinkley, south to Palmdale, and west to near Gorman. P. inornatus capture rates were typically low relative to other nocturnal rodents, and it was not found consistently at localities at which it had been captured previously. Adults were sexually dimorphic, with males significantly larger than females. P. inornatus was significantly larger than the sympatric little pocket mouse (P. longimembris) for length of head-body, total length and body mass, and possessed a relatively shorter tail. We collected 15 specimens of P. inornatus from nine localities in the western Mojave Desert for cytogenetic analysis and each possessed the same karyotype, characterized by a diploid complement of 52 chromosomes, which has not been previously described for the genus. We argue that the 52-chromosome form should be considered distinct for conservation purposes, because it occupies a relatively small geographic area that is being subjected to increasing habitat loss and fragmentation due to residential, commercial, and renewable energy development.
Key words: distribution, karyotype, longimembris group, Mojave Desert, natural history, Perognathus inornatus, San Joaquin pocket mouse
Introduction
The San Joaquin pocket mouse (Perognathus inornatus) occupies arid grassland, savanna, and desert scrub habitats in the Sacramento, San Joaquin, and Salinas Valleys and contiguous areas, the foothills of the western Sierra Nevada, the Tehachapi Mountains, and the western Mojave Desert (Best 1993; Williams et al. 1993). The species currently comprises three subspecies (inornatus, neglectus, and psammophilus). Individuals from the western Mojave Desert are similar in external and cranial characters to the subspecies neglectus, but little information is available regarding the species in the Mojave Desert (Williams et al. 1993).
Taxonomic relationships within Perognathus are based on morphological, biochemical, and cytological data (Best 1993; Williams et al. 1993). P. inornatus is part of the longimembris group, along with the little pocket mouse (P. longimembris) and Arizona pocket mouse (P. amplus) (Williams 1978). Although the genus exhibits little intraspecific karyotypic variation (Patton and Rogers 1993), P. inornatus as currently constituted includes members with more than one diploid number: P. i. inornatus (2n = 50) and P. i. neglectus (2n = 56), which inhabit the San Joaquin Valley and environs, and an undescribed population in the Sacramento Valley (2n = 60) (Slayden 1985; Best 1993; Williams et al. 1993). There is compelling evidence to suggest that P. i. inornatus and P. i. neglectus represent distinct species based in part on differing diploid numbers (Best 1993; Williams et al. 1993).
Natural communities that support P. inornatus have been converted to agricultural and urban use on a large scale in the San Joaquin Valley. As of 2000, an estimated 65% of grasslands and 64% of shrublands, habitats occupied by P. inornatus, had been lost (Kelly et al. 2005). In recent decades, the loss and fragmentation of desert scrub habitats in the western Mojave Desert resulting from development, including urban, agricultural, and renewable energy projects, has intensified. The U.S. Fish and Wildlife Service previously considered P. inornatus as a candidate for listing as threatened or endangered and the Bureau of Land Management lists it as a sensitive species (USFWS 1994; BLM 2021). Herein we provide data on distribution, morphology, karyotype, natural history, and field identification of P. inornatus from the western Mojave Desert.
Methods
Study Area
We conducted live-trapping for nocturnal small mammals at 137 locations in the western Mojave Desert between 1990 and 2017 during the course of various baseline biological investigations and environmental planning efforts. Our methods and trapping effort varied between sites and were not designed to sample the study area as a whole. Our study area included much of the Antelope Valley in portions of Kern, Los Angeles, and San Bernardino Counties. Precipitation in the western Mojave Desert is winter-dominant, occurring primarily between October and April, and varies considerably within the study area. Long-term annual precipitation averages 18.7 cm in Lancaster (Los Angeles County) in the western portion of the study area and 12.9 cm in Barstow (San Bernardino County) at the eastern edge (WRCC 2021). Winter temperatures reach 13 °C and periodic snow is not uncommon, while a marine influence keeps summer temperatures less extreme than the central and east Mojave, although highs often reach 45 °C. Most trapping locations were on bajadas with flat to gently rolling topography that typically contained multiple small, ephemeral desert washes. Vegetation communities comprised various Mojave scrub series (CNPS 2020), predominantly with creosote bush (Larrea tridentata) and saltbush scrub (Atriplex spp.) as dominant species.
Live-trapping Methods
We followed the American Society of Mammalogists guidelines regarding use of wild mammals in research (Sikes 2016). Live-trapping was conducted using Sherman™ XLK live-traps (H. B. Sherman Traps, Tallahassee, FL, USA), baited with a mixture of birdseed, rolled oats, and peanut butter in the evening and checked the following morning. Trapping intensity varied considerably between projects. We sampled 60 sites for nocturnal small mammals as part of a habitat quality analysis study on the 1,217 km2 Edwards Air Force Base. These locations were chosen at random and stratified by vegetation community, loosely based on Holland (1986) and updated for this paper according to CNPS (2020). A 100-trap grid (4 x 25 with 10-meter spacing) was sampled for three consecutive nights at each site during three separate efforts: in 1993, 2003, and on a rotating basis between 2004 and 2009. At two other sites on Edwards Air Force Base, one in creosote bush scrub and one in allscale scrub, we conducted four consecutive nights of trapping monthly between March and October 1995 using paired traps in a 12 x 12 grid with 10-m spacing. Forty-seven sites in the broader study area were sampled with 100-trap (4 x 25 or 10 x 10) grids with 10-, 25-, or 35-m spacing for three consecutive nights. These locales included six sites at the El Mirage Cooperative Management Area in San Bernardino County in 1991, three sites for the West Mojave Plan in Kern County in 1995, 18 sites at Onyx Ranch in Kern County in 2012, and 18 sites along State Route 138 in Los Angeles County in 2014 and 2015. At the remaining 28 sites, we focused live-trapping studies on detection of a diurnal species, the state-listed (Threatened) Mohave ground squirrel (Xerospermophilus mohavensis), and at these sites we left 100-trap (4 x 25 with 35-meter spacing) grids open for one night to sample for nocturnal small mammals. These locales include six sites along State Route 58 west of Hinkley San Bernardino County in 2007 and eight sites along Highway 247 south of Barstow in San Bernardino County in 2009. Overall, our sampling effort included 87,832 trap-nights.
We identified all captured small mammals to species. Individuals were marked (typically with ink) at first capture. We recorded external measurements (length of head and body, length of tail, length of hind foot, and length of ear in mm) and body mass (in g) of 228 adult P. inornatus (110 female, 118 male) and 220 adult P. longimembris (108 female; 112 male). We distinguished adult P. inornatus based on size and the ratio of the length of tail to the length of head and body (Best 1993; Williams et al. 1993). We used two-sample t-tests assuming equal variances to compare external measurements of females and males for both P. inornatus and the little pocket mouse, and between P. inornatus and little pocket mice for both females and males. We collected 15 live specimens (from nine localities) and delivered them to the Museum of Vertebrate Zoology for cytogenetic analysis.
Cytogenetic Analysis
Specimens (14 skins with skulls and one preserved in fluid) collected in 1993, 1995, and 2014 are housed at the Museum of Vertebrate Zoology (Berkeley, CA, USA; Catalog # 182266, 182346, 182539-41, 182709, 184638-43, 184679-80, and 230885). Metaphase somatic chromosomes of each specimen were processed, analyzed, and photographed following methods detailed in Patton (1967) (J. Patton, personal communication, 28 April 1993, 10 May 1995, 1 August 2014). We obtained karyotypes for all other members of the genus from Patton and Rogers (1993).
Specimen Review
We examined specimens of the longimembris group housed at the California Academy of Sciences (CAS), the Los Angeles County Museum of Natural History (LACM), the Museum of Vertebrate Zoology (MVZ), the Santa Barbara Museum of Natural History (SBMNH), and the San Diego Natural History Museum (SDNHM). We recorded measurements of intact skulls of adults with dial calipers to the nearest 0.1 mm and included the following: occipitonasal length, frontonasal length, nasal length, interorbital breadth, mastoid breadth, zygomatic breadth, length mastoid bulla, width interparietal, maxillary toothrow, and length of maxillary diastema (as defined in Hoffmeister 1986).
Results
Live-trapping Data
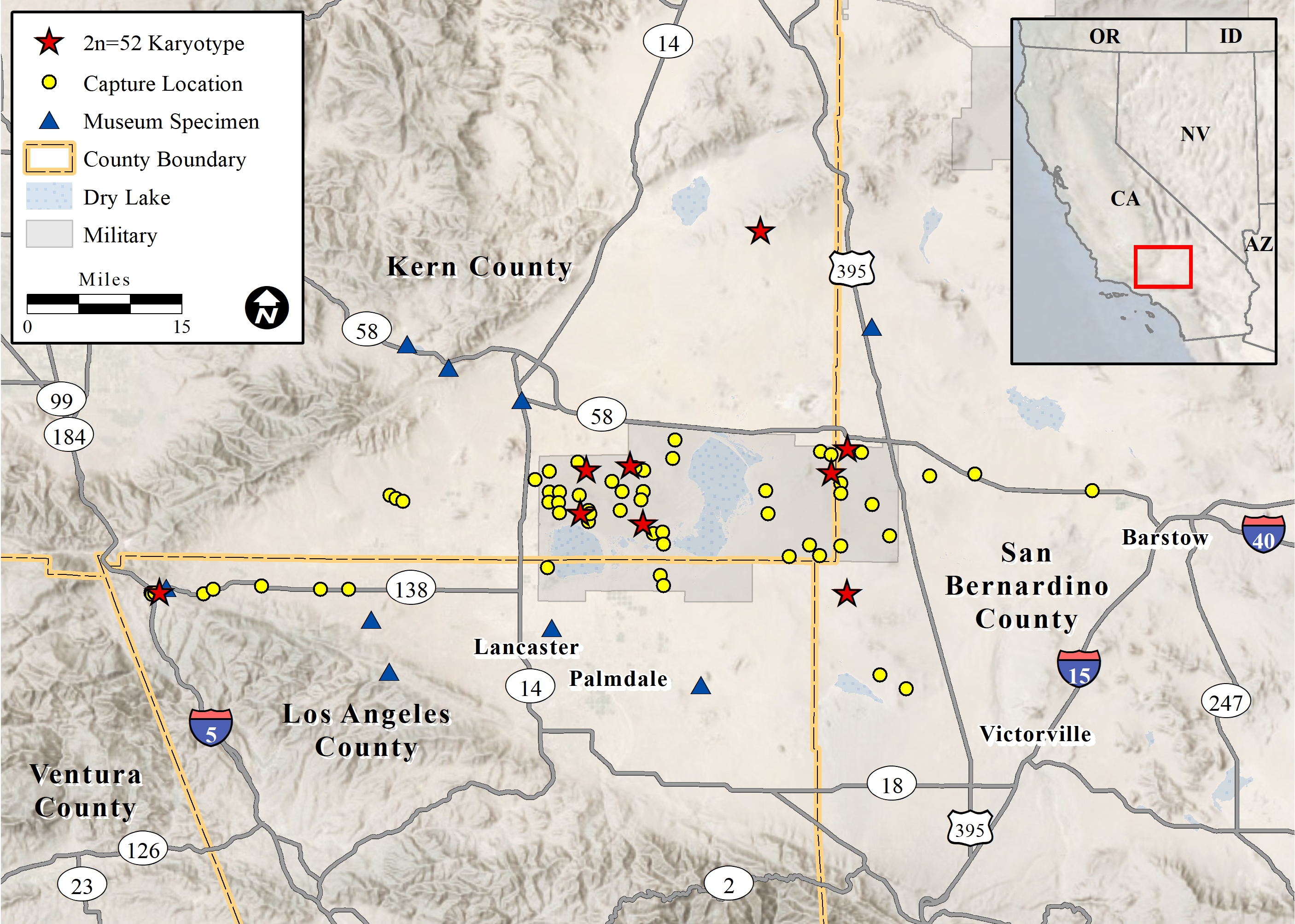
We captured P. inornatus in the western Mojave Desert at elevations from 668–1,109 m above mean sea level (Fig. 1). Habitats at occupied sites included creosote bush scrub, allscale scrub, Joshua tree woodland, rubber rabbitbrush scrub, spinescale scrub, and California juniper woodland (CNPS 2020). Shrub canopy was typically intermittent to open (Fig. 2). We found the species most frequently on well-drained, granite-derived soils and did not capture it in rocky substrates or in low-lying, poorly-drained soils.

We trapped the following rodents in association with P. inornatus: long-tailed pocket mouse (Chaetodipus formosus), little pocket mouse, Merriam’s kangaroo rat (Dipodomys merriami), Great Basin kangaroo rat (D. microps), Panamint kangaroo rat (D. panamintinus), desert woodrat (Neotoma lepida), southern grasshopper mouse (Onychomys torridus), North American deermouse (Peromyscus maniculatus), western harvest mouse (Reithrodontomys megalotis), white-tailed antelope squirrel (Ammospermophilus leucurus), and Mohave ground squirrel (Xerospermophilus mohavensis). P. inornatus and P. longimembris co-occurred at 33 of 137 sites sampled (24%).
At the 60 locations on Edwards Air Force Base sampled on three occasions over the course of 16 years, P. inornatus was captured at 23 sites (38.3%) in 1993, 21 sites (35.0%) in 2003, and 31 sites (51.7%) between 2004 and 2009. It was detected during all three trapping efforts at only nine sites (15.0%) and on only one of three occasions at 18 sites (30.0%).
P. inornatus was captured infrequently relative to other nocturnal small mammals. At Edwards Air Force Base, the species comprised 79 of 6,352 individuals (1.2%) captured in 1993, 55 of 1,942 individuals (2.8%) in 2003, and 185 of 4,407 individuals (4.2%) between 2004 and 2009. In comparison, P. longimembris accounted for 6.7%, 13.1% and 8.3% of nocturnal small mammals in 1993, 2003, and 2004-2009, respectively. At 107 sites, sampled with a 100-trap grid for three consecutive nights, the capture rate (number of individuals per 100 trap-nights) of P. inornatus was 0.57, compared with 1.83 for P. longimembris, and 24.70 for all nocturnal small mammals. However, P. inornatus was sometimes abundant on individual grids, and the capture rate was as high as 8.33 individuals per 100 trap-nights. More intensive trapping also resulted in increased detection of the species. On two 144-station grids trapped monthly between March and October using paired traps, P. inornatus comprised 22.4 % and 15.2% of all nocturnal small mammals captured, respectively.
During monthly trapping between March and October, only adult males were captured in March. Females were in reproductive condition in April, May, and June. Juveniles were captured between May and August. Capture rates peaked in August, before dropping dramatically in September, and only a single individual was captured in October. The average distance between successive captures and home range size were considerably larger for adult males than females.
Morphology
P. inornatus from the Mojave Desert closely resembled P. i. neglectus from the western San Joaquin Valley and Inner Coast Ranges. The dorsal coloration was typically ochraceous to buff overlaid with black (Fig. 3). The pelage was soft and without bristles (guard hairs) on the rump. The ventral coloration was whitish. The lateral line was fulvous and typically distinct. Subauricular spots were distinct and buffy to white. There was a faint to obvious black crescent on the nose. The feet were white, and the soles had sparse hairs. The tail was white or slightly bicolored, penciled, and without a crest. The tail was slightly longer than the length of the head and body (Williams et al. 1993). Many individuals, primarily adult males, emitted a distinctive “buttered popcorn” odor, which was not detected in any of the P. longimembris that we captured.

P. inornatus males were significantly larger than females for length of head and body, length of tail, length of hind foot, and body mass (a = 0.05), although there was broad overlap in measurements (Table 1). In contrast, there were no significant differences between external measurements of males and females in P. longimembris. Both male and female P. inornatus were significantly larger than P. longimembris in total length, length of head and body, length of hind foot, and body mass (a = 0.05), while males were also significantly larger in length of tail. (Table 1; Fig. 4). For P. inornatus, body mass ranged between 8 and 24 grams and averaged 15.7 grams (n = 228), while for P. longimembris body mass ranged between 6 and 13 grams and averaged 9.5 grams (n = 220). In P. inornatus, the ratio of the length of tail to the length of the head and body averaged 1.06 (n = 228), whereas in P. longimembris the ratio averaged 1.21 (n = 220).
Table 1. Descriptive statistics of external measurements of adult Perognathus inornatus and P. longimembris from the Mojave Desert, CA, USA. (P. inornatus female: n = 110 and male: n = 118; P. longimembris female: n = 108 and male: n = 112)
| Species | Sex | Statistic | Total length (mm) | Length of head-body (mm) | Length of tail (mm) | Length of hind foot (mm) | Body mass (g) | Head-body/tail |
|---|---|---|---|---|---|---|---|---|
| P. inornatus | Female | mean | 142.58 | 69.16 | 73.50 | 19.69 | 14.62 | 1.06 |
| P. inornatus | Female | s | 7.90 | 4.23 | 4.56 | 1.06 | 2.52 | 0.06 |
| P. inornatus | Female | min | 124 | 60 | 62 | 16 | 9 | 0.91 |
| P. inornatus | Female | max | 164 | 80 | 88 | 22 | 23 | 1.17 |
| P. inornatus | Male | mean | 149.51 | 72.60 | 76.95 | 20.15 | 16.58 | 1.06 |
| P. inornatus | Male | s | 8.74 | 4.69 | 4.85 | 1.00 | 2.78 | 0.05 |
| P. inornatus | Male | min | 127 | 60 | 63 | 17 | 11 | 0.90 |
| P. inornatus | Male | max | 171 | 85 | 88 | 23 | 24 | 1.18 |
| P. longimembris | Female | mean | 132.97 | 60.20 | 72.86 | 18.57 | 9.70 | 1.21 |
| P. longimembris | Female | s | 6.79 | 3.88 | 4.52 | 0.86 | 1.63 | 0.09 |
| P. longimembris | Female | min | 117 | 51 | 61 | 17 | 7 | 1.05 |
| P. longimembris | Female | max | 155 | 71 | 85 | 20 | 13 | 1.48 |
| P. longimembris | Male | mean | 133.38 | 60.43 | 72.85 | 18.47 | 9.28 | 1.20 |
| P. longimembris | Male | s | 7.62 | 4.13 | 4.65 | 1.08 | 1.43 | 0.08 |
| P. longimembris | Male | min | 116 | 52 | 61 | 16 | 6 | 1.05 |
| P. longimembris | Male | max | 152 | 71 | 82 | 21 | 13 | 1.42 |

Skulls of P. inornatus from the Mojave Desert closely resemble those of P. i. neglectus. Descriptive statistics of cranial measurements are presented in Table 2. The interorbital breadth was relatively narrow and averaged 22% of the occipitonasal length. The mastoid bullae were moderately inflated, nearly meeting anterior-ventrally, and with the posterior end extending beyond the plane of the occiput.
Table 2. Descriptive statistics of cranial measurements of 2n = 52 P. inornatus (7 females and 7 males). All measurements are in mm.
| Sex | Statistic | Occipitonasal length | Frontonasal length | Nasal length | Interorbital breadth | Mastoid breadth | Zygomatic breadth | Length mastoid bulla | Width interparietal | Maxillary toothrow | Maxillary diastema |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Females | mean | 23.23 | 15.83 | 8.60 | 5.01 | 13.13 | 12.19 | 8.06 | 4.24 | 3.26 | 6.13 |
| Females | s | 0.59 | 0.44 | 0.22 | 0.17 | 0.25 | 0.55 | 0.33 | 0.42 | 0.15 | 0.25 |
| Females | min | 22.5 | 15.2 | 8.4 | 4.9 | 12.8 | 11.7 | 7.6 | 3.9 | 3.1 | 5.7 |
| Females | max | 24.3 | 16.5 | 9.0 | 5.3 | 13.5 | 13.2 | 8.5 | 5.1 | 3.5 | 6.5 |
| Males | mean | 23.87 | 16.34 | 8.99 | 5.34 | 13.19 | 12.33 | 8.21 | 4.26 | 3.17 | 6.40 |
| Males | s | 0.87 | 0.67 | 0.61 | 0.15 | 0.27 | 0.42 | 0.33 | 0.58 | 0.18 | 0.31 |
| Males | min | 22.7 | 15.3 | 8.4 | 5.1 | 12.8 | 11.8 | 7.7 | 3.7 | 2.8 | 6.0 |
| Males | max | 24.9 | 16.9 | 9.9 | 5.6 | 13.6 | 13.1 | 8.6 | 5.4 | 3.3 | 6.7 |
Cytogenetic Analysis
All 15 P. inornatus examined from nine localities in the Mojave Desert had the same karyotype, characterized by a diploid number of 52 and a fundamental number (number of autosomal arms) of 88 (J. Patton, personal communication, 28 April 1993, 10 May 1995, 1 August 2014). Nineteen chromosome pairs were bi-armed, and six pairs were acrocentric. One pair of bi-armed chromosomes possessed terminal satellites. The X chromosome was sub-metacentric, and the Y chromosome was metacentric. The diploid number (2n = 52) differed from all other members of the longimembris species group and from all other members of the genus (Patton and Rogers 1993). The 2n = 52 karyotype possessed a single pair of satellite-bearing, biarmed autosomes while both P. i. inornatus and P. i. neglectus have two pairs with satellites (Williams 1978). P. inornatus collected from near Monolith (Kern County) in 2015 also possessed the same 2n = 52 karyotype (J. Patton, personal communication, 15 February 2016).
Specimen Review
Museum specimens of P. inornatus from the Mojave Desert included: Kern Co.: LACM 3136, adult male, Mojave (labeled as P. i. neglectus); MVZ 126001, adult male, 0.8 km south Cameron (labeled as P. i. inornatus); MVZ 192765-74, Monolith, 12.9 km east Tehachapi (labeled as P. i. neglectus); Los Angeles Co.: LACM 4368-71, 1.6 km west Quail Lake (labeled as P. i. neglectus); LACM 5021-26, Elizabeth Lake (labeled as P. i. neglectus); MVZ 44317, adult male, 1.6 km south Peck’s [=Piute] Butte (labeled as P. longimembris, but with a note that it may be P. inornatus); SBMNH 57-51, adult male, Fairmont (labeled as P. i. neglectus); San Bernardino Co.: LACM 047005, adult male, 7.2 km east, 16.9 km north Boron (labeled as P. i. inornatus) (Fig. 1). Three P. inornatus specimens in poor condition from Los Angeles County were not included in Fig. 1: LACM 3137, Palmdale; LACM 5027, Lake Hughes; and LACM 5028, Gorman.
All P. inornatus specimens from locations in the Tehachapi Mountains and southern Sierra Nevada were labeled P. i. inornatus and were similar in appearance to 2n=50 specimens from the east side of the San Joaquin Valley. These included Kern County: LACM 86783-4, Chimney Creek; LACM 89241-46, 4 km north, 2.4 km east Twin Oaks; LACM 60065-66 southwest Walker Basin; MVZ 16191, Kern River at Bodfish; MVZ 16197-16203, Kern River at Isabella; MVZ 60065-6, southwest Walker Basin.
Discussion
The San Joaquin pocket mouse is known from the western Mojave Desert based on museum records dating back to the 1930s (Best 1993; Williams et al. 1993; Wilson and Ruff 1999). However, the region is not included in published range maps (Ingles 1965; Hall 1981; Jameson and Peeters 1988; Best 1993; Wilson and Ruff 1999; Kays and Wilson 2002). Little information is available regarding the species in the west Mojave Desert, possibly due to its low relative abundance and seasonal inactivity.
Capture locations reported herein, together with museum specimens, indicate that, at a minimum, P. inornatus ranges north to the Rand Mountains, east to the vicinity of Hinkley, south to Palmdale, and west to near Gorman. These locales are largely within the Antelope Valley basin covering an area of approximately 7,000 km2. In general, the known range of P. inornatus in the Mojave Desert coincides with a habitat map generated by the United States Geological Survey Gap Analysis Project (USGS 2018). Additional research is necessary to refine the limits of its range, especially in the east and north.
P. inornatus were captured infrequently relative to other nocturnal rodents and were not consistently captured at sites at which they had been previously found. The species may undergo dramatic year-to-year variation in abundance, which has been documented in P. inornatus from the San Joaquin Valley (Cypher 2001; Germano and Saslaw 2017). Our results, using intensive trapping methods (monthly, four-night sessions between March and October with paired traps) suggest that relative abundance may be underestimated using lower-intensity trapping efforts. High capture rates of more common species, for example, may preclude P. inornatus from being detected at some locales during certain years.
Results from monthly trapping indicated that P. inornatus was seasonally inactive and likely hibernated in the fall and winter, as reported for other members of the species group (Kenagy 1973; Best 1993; Wilson and Ruff 1999). In P. longimembris, the duration of winter inactivity is dependent on the availability of food resources (Kenagy 1973).
P. inornatus co-occurred with P. longimembris at 24% of all sites sampled. Distinguishing between the two species typically requires examination of adult individuals, given their similar external characters (Best 1993). Although there is some overlap in measurements, adult P. inornatus can be reliably distinguished from adult P. longimembris based on larger size and a relatively shorter tail. Body mass of P. inornatus averaged 15.6 g (range 9–24 g), while P. longimembris averaged 9.5 g (range: 6–13g). The ratio of length of tail to length of head and body averaged 1.06 (range: 0.90–1.18) in P. inornatus and 1.21 (range: 1.05–1.48) in P. longimembris. Because males are larger than females in P. inornatus but not in P. longimembris, there is more overlap in measurements between the two species for females. Although not detectable in all individuals, we believe the distinctive “buttered popcorn” odor is diagnostic for P. inornatus. This odor was assumed to originate from sebaceous caudal glands as described by Quay (1965) for other Perognathus species including the Arizona pocket mouse (Wilson and Ruff 1999) and may be used for olfactory communication or territorial marking.
Taxonomic relationships among the three currently recognized subspecies of P. inornatus (P. i. inornatus, neglectus, and psammophilus) are unresolved and it is likely that more than one species is included within the group (Williams et al. 1993). The 50-chromosome karyotype characterizing P. i. inornatus is known from the floor of the San Joaquin Valley, western foothills of the Sierra Nevada, and Tehachapi Mountains. The 56-chromosome karyotype associated with P. i. neglectus is generally larger than P. i. inornatus and has been identified from the western side of the San Joaquin Valley and the Carrizo Plain (Patton and Rogers 1993). P. i. psammophilus is known from the Salinas Valley and may be a synonym of P. i. neglectus (Best 1993; Williams et al. 1993). A single specimen collected from Lake County near the northern limits of the species’ range had a diploid number of 60 (Slayden 1985; Williams et al. 1993), indicating that there may be additional cytogenetic variation within the group. In our study, P. inornatus collected from nine widely separated locations in the western Mojave Desert all possessed the same karyotype with a diploid number of 52. This diploid number is not found elsewhere in the genus (Patton and Rogers 1993). The consistency with which the karyotype was identified establishes that it is widespread in the region and not an aberrant form.
Although 2n = 52 P. inornatus from the Mojave Desert and 2n = 56 P. inornatus from the western San Joaquin Valley are phenotypically similar, close morphological similarity between reproductively distinct species has been demonstrated elsewhere in the genus (Lee and Engstrom 1991). The Tehachapi Mountains and southern Sierra Nevada present a geographic barrier between P. inornatus from the Mojave Desert and P. i. neglectus from the San Joaquin Valley. The significance of the Tehachapi Mountains in the biogeography of other heteromyids has been discussed (Stock 1974; Schmidly et al. 1993).
Much of the range of P. inornatus in the western Mojave Desert is relatively undeveloped. However, development, including agricultural and renewable energy, particularly in the western Antelope Valley, has increased dramatically. Our results show that P. inornatus from the western Mojave Desert are cytogenetically distinct from all other members of the genus. Resolution of the taxonomic relationships among the karyotypes that are included within P. inornatus is critical to determining conservation priorities. Until a comprehensive taxonomic review is conducted, we feel it is important to consider the 52-chromosome form independently for conservation purposes, given that it occurs at relatively low frequencies and occupies a small geographic area that is being subjected to increasing habitat loss and fragmentation as the result of development.
Acknowledgments
We extend our deepest thanks to J. Patton, whose advice, encouragement, and technical assistance made this paper possible. J. Patton processed, analyzed, and photographed metaphase somatic chromosomes of each specimen. We thank the following for providing access to the mammal collections examined: K. Cebra (CAS), L. Barkley and D. Janeger (LACM), J. Patton (MVZ), P. Collins (SBMNH), and P. Unitt (SDNHM). Data from Edwards Air Force Base in the early 1990s were gathered under contract to Tetra Tech, Inc., in support of their environmental analysis contract with GRW Engineers, Inc., and the United States Army Corps of Engineers, Sacramento District for the Air Force Flight Test Center, Environmental Management Office, Edwards Air Force Base, California (Contract # DCA05-C-91-0130), for the implementation of the Installation Restoration Program and, in subsequent years in support of the goals of the base’s Integrated Natural Resources Management Plan. Trapping at the El Mirage Cooperative Management Area was conducted under contract to the Bureau of Land Management (Contract # B950C10026). Trapping for the West Mojave Plan was conducted under contract to Cal Poly Pomona Foundation, Inc. (Contract # FG3619). Trapping for the State Route 58 Realignment and State Route 138 Northwest Corridor Improvement Projects were conducted under contract to Caltrans (Contracts 08A1579 and 07A2269, respectively). We thank J. (Sawasaki) Fraser, K. Glinka, B. Haley, S. Higgins, S. Montgomery, C. Murphy, K. Simon, C. Steiner, K. Wasz, and P. Wasz for field assistance. P. Brylski, R. Condit, and J. Patton provided comments that improved earlier versions of the manuscript. Thanks to J. Corona-Bennett for assistance updating vegetation community classifications and M. Guidry for improving the range map.
Literature Cited
- Best, T. L. 1993. Perognathus inornatus. Mammalian Species 450:1–5.
- California Native Plant Society (CNPS). 2020. A manual of California vegetation, online edition. Available from: http://www.cnps.org/cnps/vegetation (13 December 2020).
- Cypher, B. L. 2001. Spatiotemporal variation in rodent abundance in the San Joaquin Valley, California. Southwestern Naturalist 46:66–75.
- Germano, D. J., and L. R. Saslaw. 2017. Rodent community dynamics as mediated by environment and competition in the San Joaquin Desert. Journal of Mammalogy 98:1615–1626.
- Hall, E. R. 1981. The Mammals of North America. 2nd edition. John Wiley & Sons, New York, NY, USA.
- Hoffmeister, D. F. 1986. Mammals of Arizona. University of Arizona Press, Tucson, AZ, USA.
- Holland, R. F. 1986. Preliminary descriptions of the terrestrial natural communities of California. State of California, The Resources Agency, Department of Fish and Game, Sacramento, CA, USA.
- Ingles, L. G. 1965. Mammals of the Pacific States. Stanford University Press, Redwood City, CA, USA.
- Jameson, E. W., Jr., and H. J. Peeters. 1988. California Mammals. University of California Press, Berkeley, CA, USA.
- Kays, R. W., and D. E. Wilson. 2002. Mammals of North America. Princeton University Press, Princeton, NJ, USA.
- Kelly, P. A., S. E. Phillips, and D. F. Williams. 2005. Documenting ecological change in time and space: the San Joaquin Valley of California. Pages 57–78 in E. A. Lacey and P. Myers, editors. Mammalian Diversification: From Chromosomes to Phylogeography. Publications in Zoology Series, University of California Press, Berkeley, CA, USA.
- Kenagy, G. J. 1973. Daily and seasonal patterns of activity and energetics in a heteromyid rodent community. Ecology 54(6):1201–1219.
- Lee, T. E., Jr., and M. D. Engstrom. 1991. Genetic variation in the silky pocket mouse (Perognathus flavus) in Texas and New Mexico. Journal of Mammalogy 72:273–285.
- Patton, J. L. 1967. Chromosome studies of certain pocket mice, genus Perognathus (Rodentia: Heteromyidae). Journal of Mammalogy 48:27–37.
- Patton, J. L., and D. S. Rogers. 1993. Cytogenetics. Pages 236–258 in H. H. Genoways and J. H. Brown, editors. Biology of the Heteromyidae. Special Publication 10. American Society of Mammalogists.
- Quay, W. B. 1965. Variation and taxonomic significance in the sebaceous caudal glands of pocket mice (Rodentia:Heteromyidae). Southwestern Naturalist 10(4):282–287.
- Schmidly, D. J., K. T. Wilkins, and J. N. Derr. 1993. Biogeography. Pages 319–356 in Biology of the Heteromyidae (H.H. Genoways and J. H. Brown, eds). Special Publication 10, American Society of Mammalogists, Topeka, KS, USA.
- Sikes, R. S., and the Animal Care and Use Committee of the American Society of Mammalogists. 2016. 2016 Guidelines of the American Society of Mammalogists for the use of wild mammals in research and education. Journal of Mammalogy 97:663–688.
- Slayden, O. D. 1985. A record of the San Joaquin pocket mouse Perognathus inornatus, from Lake County, California. Southwestern Naturalist 30(3):466.
- Stock, A. D. 1974. Chromosome evolution in the genus Dipodomys and its taxonomic and phylogenetic implications. Journal of Mammalogy 55:505–526.
- U.S. Department of the Interior, Bureau of Land Management (BLM). 2021. California Threatened and Endangered Species. Available from: https://www.blm.gov/programs/fish-and-wildlife/threatened-and-endangered/state-te-data/California
- U.S. Geological Survey (USGS) – Gap Analysis Project. 2018. San Joaquin pocket mouse (Perognathus inornatus) mSJPMx_CONUS_2001v1 habitat map: U.S. Geological Survey data release. Available from: https://doi.org/10.5066/F780511V
- U.S. Fish and Wildlife Service (USFWS). 1994. Endangered and threatened wildlife and plants; animal candidate review for listing as endangered of threatened species; proposed rule. Federal Register 59:58982–59028.
- Williams, D. F. 1978. Karyological affinities of the species groups of silky pocket mice (Rodentia: Heteromyidae). Journal of Mammalogy 59:599–612.
- Williams, D. F., H. H. Genoways, and J. K. Brown. 1993. Taxonomy. Pages 38–196 in H. H. Genoways and J. H. Brown, editors. Biology of the Heteromyidae. Special Publication 10. American Society of Mammalogists, Topeka, KS, USA.
- Wilson, D., and S. Ruff. 1999. The Smithsonian Book of North American Mammals. Smithsonian Institution Press, Washington, D.C., USA.
- Western Regional Climate Center (WRCC). 2020. California climate summaries (1974–2016). Available from: http://www.wrcc.dri.edu/summary/climzmaz.html (November 2020).

