FULL RESEARCH ARTICLE
Erica Donnelly-Greenan1,2, Corinne Gibble3*, and Jessie Beck2
1 Save Our Shores, 345 Lake Avenue #A, Santa Cruz, CA 95062, USA (current affiliation)
2 Oikonos Ecosystem Knowledge, P.O. Box 2570, Santa Cruz, CA 95062, USA
3 California Department of Fish and Wildlife, Office of Spill Prevention and Response, 151 McAllister Way, Santa Cruz, CA 95060, USA
*Corresponding Author: corinne.gibble@wildlife.ca.gov
Published 10 July 2023 • doi.org/10.51492/cfwj.109.5
Abstract
Examinations of seabirds from oil spills provide resource managers demographic and trauma related data that inform damage assessments for affected species. We examined black skimmers (Rynchops niger; n = 49) from the Deepwater Horizon oil spill for demographics, ingested petroleum, and plastics. The majority were hatch-year males in poor body condition (57%), with atrophied, dehydrated, or pale organs possibly attributed to external oil exposure. Six (12%) black skimmers had evidence of external oiling, three (6%) were fouled by crude oil, one (2%) had biological fouling, and two (4%) were fouled in a synthetic substance, suspected to be chemical dispersant. Damage to organs suggested that internal examinations should be performed alongside external surveys to provide a thorough understanding of the total number of individuals impacted by a spill, and that timely internal examinations are essential. No evidence of ingested petroleum or plastics was found.
Key words: black skimmers, Deepwater Horizon oil spill, demographics, oil ingestion, oil spill, plastic ingestion, Rynchops niger, seabirds
| Citation: Donnelly-Greenan, E., C. Gibble, and J. Beck. 2023. Assessing impacts of oil and ingested plastic in black skimmers from the Deepwater Horizon oil spill. California Fish and Wildlife Journal 109:e5. |
| Editor: Samreen Siddiqui, Bay Delta Region |
| Submitted: 15 February 2023; Accepted: 27 March 2023 |
| Copyright: ©2023, Donnelly-Greenan et al. This is an open access article and is considered public domain. Users have the right to read, download, copy, distribute, print, search, or link to the full texts of articles in this journal, crawl them for indexing, pass them as data to software, or use them for any other lawful purpose, provided the authors and the California Department of Fish and Wildlife are acknowledged. |
| Funding: This project was funded by the California Department of Fish and Wildlife Oil Spill Response Trust Fund through the Oiled Wildlife Care Network at the Wildlife Health Center, School of Veterinary Medicine, University of California, Davis, Research Agreement Number 201703366-04. |
| Competing Interests: The authors have not declared any competing interests. |
Introduction
In 2010, the Deepwater Horizon (DWH) wellhead blowout and oil spill expelled approximately 3.19 million barrels (134 million gallons) of crude oil into the Gulf of Mexico (Beyer et al. 2016), contaminating the ocean floor, the surrounding water column, adjacent coastal environments, and exposing large numbers of marine species (Beyer et al. 2016). The consequences of the spill included dramatic impacts to marine and coastal wildlife, including multiple species of seabirds (Beyer et al. 2016; Deepwater Horizon Natural Resource Damage Assessment Trustees 2016). For seabirds, exposure to crude oil and associated polycyclic aromatic hydrocarbons (PAHs) can cause hypothermia, organ damage, decreased body mass, hemolytic anemia, and immunosuppression (Leighton 1993; Briggs et al.1997; Paruk et al. 2016; Bursian et al. 2017). Negative health effects are not limited to heavily oiled birds; studies have shown oxidative injury to erythrocytes, erythrocytic inclusion bodies, increased reticulocytes, and reduced packed cell volumes in birds without external visible oiling (Fallon et al. 2017). Additionally, some dispersants and chemical solvents used during spill clean-up can have sublethal impacts on seabirds (Peakall and Mackay 1987) and can negatively impact seabird feather structure and thermoregulatory functions (Jenssen and Ekker 1990; Whitmer et al. 2018).
During oil spills, seabirds can be exposed to oil both externally and internally (Vauk et al. 1989; Fallon et al. 2017; Whitmer et al. 2018). While oil spill responders and wildlife rehabilitators routinely quantify external effects of oil, documenting the internal effects of oil exposure is logistically more difficult. However, systematic necropsies of seabird carcasses (during and after spills) can enhance the understanding of the range of impacts on seabirds (Vauk et al. 1989; Balserio et al. 2005). This may be especially true in cases of little to no external fouling (Vauk et al. 1989). Additionally, post-mortem examinations may assist rehabilitators and veterinarians in improving care during the oil spill and assist in management planning for future disasters (Whitmer et al. 2018).
Black skimmers (Rynchops niger) are one of many seabird species that were impacted by DWH (USFWS 2011). This species occupies the Gulf of Mexico and the California coast, and utilizes coastal habitats for foraging, nesting, and over-wintering (Furfey 2014). Black skimmers are tactile foragers that fly low over the surface water, submerging their lower mandible, and skimming the water for fish, insects, crustaceans, and mollusks (Furfey 2014). Due to their unique foraging strategy, preening behavior, and coastal habitat use, we predicted that black skimmers within proximity of DWH may have been at greater risk for ingesting oil.
Oil ingestion could be particularly hard to document if it is grossly confused with melena (digested blood) due to similarity in dark color and sometimes gritty consistency (Wilson 1990). If there is evidence of oil in the gastrointestinal track (GI), possible damages include physiological injury to gastrointestinal tissues, grossly apparent gastrointestinal lesions, and damage to liver and renal tissues (Szaro et al. 1978). Previous studies have also documented severe dehydration and emaciation (Khan and Ryan 1991; Balseiro et al. 2005). To correctly identify the range of affects oil exposure has on seabirds, occult blood tests can be used to help distinguish between melena and ingested oil (M. Miller, California Department of Fish and Wildlife, pers. comm.).
For black skimmers, ingestion of other petroleum-based products, such as plastics, may be an additional concern due to their surface-skimming foraging strategy. Pre- and post-consumer plastics that gather in surface waters can be mistaken for prey species based on olfactory signals (Savoca et al. 2016), and ingested incidentally during foraging (Sileo et al. 1989), or ingested secondarily through their prey (Hammer et al. 2016). In other surface feeding seabirds, including other species within the Laridae family, individuals can experience sublethal and lethal impacts from ingested plastics or associated pollutants (Spear et al. 1995; Rapp et al. 2017; Tanaka et al. 2019; Kühn and van Franeker 2020). Plastics may similarly affect black skimmers if they ingest them; however, published studies investigating plastic ingestion in black skimmers are sparse (Kühn and van Franeker 2020). Further research on the health ramifications related to both forms of petroleum ingestion (oil and plastics) may help guide treatment in rehabilitation facilities, and inform best achievable care for oiled seabirds.
Herein, we examine black skimmers collected during wildlife response to DWH for ingested petroleum and plastics. We additionally present ancillary demographic (age, sex) and ecological (body condition) information, and report findings collected during gross necropsies.
Methods
Collection, Necropsy, and Demography
Black skimmer carcasses (n = 45) were collected, in varying stages of physical fouling and decomposition, in the field (between 29.10974, –90.03756 and 30.39298, –88.92823) between June and October of 2010 during the DWH response. Four birds were collected live and brought to Theodore Wildlife Rehabilitation Center, one of four wildlife rehabilitation centers established to support the oil spill response. All four died prior to oil removal [one individual was determined to be dead on arrival, one euthanized, and two died in-care (one within 48 hrs and the other within one week)]. All carcasses were stored frozen (–20˚C until examination; 2010–2017).
We defrosted and examined carcasses in 2017 via systematic necropsy to investigate the presence and potential impacts of petroleum, at the California Department of Fish and Wildlife, Office of Spill Prevention and Response, Marine Wildlife Veterinary Care and Research Center, Santa Cruz (CDFW-MWVCRC). Internal examinations were conducted following necropsy protocols derived from van Franeker (2004) and CDFW-MWVCRC programs. Post-mortem investigations included documentation of demographics (age, class, and sex distribution), morphometrics, percentage of external fouling, molt, lesions, and gross health of organs to indicate prevalence of disease. We determined age by examination of plumage (Pyle 2009), and the presence/absence and size of bursa of Fabricius, (herein bursa), which is only present in juvenile birds (Broughton 1994), and gonadal development. To assign age classification to the individuals with decomposed gonads, we used plumage characteristics. For black skimmers, differentiating between adult and hatch year age classification is straightforward as the plumage differences are very distinguishable (Pyle 2009). We collected complete gastrointestinal tracts including proventriculus and ventriculus for investigation of oil and plastic ingestion following van Franeker (2004).
External and Internal Examination
We estimated and recorded percentage of oil fouling, plumage and feather molt status, and morphometric measurements including wing chord, tarsus, and culmen. The status of feather molt was scored for the primary feathers and the combination of wing, body, head, and tail molt statuses in conjunction with plumage coloration assisted to distinguish between adults and immatures (Pyle 2009).
Condition indices for pectoral muscle, subcutaneous, and adipose/intestinal fat stores, as well as gonad scores for aging were adapted from van Franeker (2004). To quantify internal body condition the pectoralis-supracoracoideus (breast) muscle complex was scored from 0 (severely emaciated: muscle significantly below keel-line) to 3 (excellent body condition: muscle at or above keel-line) and subcutaneous and adipose fat stores were scored from 0 (no fat) to 3 (very fat; van Franeker 2004). The codes for the pectoralis muscle complex and the fat stores were combined to generate an inclusive body condition (condition index) score for each bird by summing the scores from the breast muscle complex and subcutaneous fat with a maximum score of six. Inclusive body conditions were grouped such that code of 0–2 was ‘poor’ condition, 3–4 was ‘moderate’ condition, and 5–6 was ‘good’ condition. Birds without breast muscles and a subcutaneous score (due to decomposition or scavenging) were labeled as unknown body condition. We examined other major organs (heart, lungs, kidneys, and liver) and took notes/photographs if an abnormal appearance was apparent (aside from changes in organ appearance as a product of freezer artifact). Extremely scavenged or decomposed carcasses were not assigned tissue health scores. Gonads were examined and measured to determine sex and reproductive maturity of each bird. For males, testis length and width were measured (mm). For females, we measured ovary length and width, and the diameter of the largest ovarian follicle (mm). An oviduct development score (1 to 4 from juvenile to breeding adult) was assigned. To further aid in estimations of maturity, we noted the presence/absence and size (if present) of bursas and recorded the presence/absence of a brood patch. If the bursa was present, the length and width were measured. It was assumed that only juvenile birds contained apparent to prominent bursas (Broughton 1994; van Franeker 2004).
Gastrointestinal Processing, Testing, and Sorting
During necropsy, we severed the proventriculus and ventriculus portion of the GI above the proventriculus and below the ventriculus and stored frozen to assess ingested plastics and oil. The remaining portion of the GI for each bird was opened along its length, and the presence/absence of suspected ingested oil was recorded. If melena and other dark-pigmented materials were detected, we conducted further testing to evaluate the presence of petroleum-based products. Melena was confirmed (with control tests) by using occult blood test panels (Cenogenics Tri-slide stool blood test, Cenogenics Corporation, Morganville, NJ, USA). This technique is a widely used diagnostic test validated by the medical community for human use and has been used by veterinarians to diagnose blood in stool of domestic and laboratory animals (Gilson et al. 1990; Valero et al. 2008). Here, we apply Tri-Slide™ Three-Section Slide Kits (two test sites, a negative, and a positive control; http://www.cenogenics.net/stool-occult-blood.html) panels to the black skimmer GI contents, which may be the first time this test has been used on seabirds. The occult blood test panels could rule out the presence of melena, but could not alone confirm or deny the presence of petroleum. If petroleum was suspected, we noted the odor and material consistency, particularly if properties of crude oil (dark reddish-brown and possibly aromatic) were apparent (Deepwater Horizon Natural Resource Assessment Trustees 2016). Stomach chambers (proventriculus and ventriculus) were later thawed and processed separately to account for potentially different residence times of stomach contents. We collected all stomach chamber contents in a 0.07 mm mesh sieve and were rinsed to remove soft prey tissues. Stomach contents were then sorted under a binocular microscope and categorized by type following van Franeker (2004).
Results
Necropsy and Demography
Of the 49 black skimmers examined, the majority (69%, 34/49) were in poor body condition, 27% (14/49) were in moderate condition, 2% (1/49) were in good condition, and 2% (1/49) were assigned as unknown condition due to advanced decomposition. There were a greater number of males (65%, 32/49) than females (24%, 12/49) and most individuals were of hatch year age (84%, 41/49) versus adult age (16%, 8/49). No immature birds older than hatch year were documented. Five birds (10%, 5/49) were of unknown sex, due to advanced body decomposition (Table 1). Morphometrics were evaluated for reference and to contribute to baseline information for this species (Table 2).
Table 1. Body condition indices of examined black skimmers (n= 49) as defined by metrics established by van Franeker (2004), where scores 0–1 are considered poor body condition, scores 3–4 are considered moderate body condition, and scores 5–6 are considered to be good body condition. One bird was considered to be unknown in body condition score due to state of decomposition.
| Sex/Age | 0 | 1 | 2 | 3 | 4 | 5* | 6* | Unknown |
| Female | 2 | 3 | 3 | 1 | 3 | – | – | – |
| Adult (F) | – | 1 | 1 | – | 1 | – | – | – |
| Hatch Year (F) | 2 | 2 | 2 | 1 | 2 | – | – | – |
| Male | 13 | 7 | 2 | 7 | 2 | – | 1 | – |
| Adult (M) | – | 2 | – | 2 | – | – | – | – |
| Hatch Year (M) | 13 | 5 | 2 | 5 | 2 | – | 1 | – |
| Adult (Unknown) | 1 | – | – | – | – | – | – | – |
| Hatch Year (Unknown) | 2 | – | 1 | – | – | – | – | 1 |
| Total | 18 | 10 | 6 | 8 | 5 | 0 | 1 | 1 |
Table 2. Morphometric measurements of examined black skimmers (n = 44) from the Deepwater Horizon Oil Spill, including average, range of measurements, and standard deviation of the upper culmen (mm), tarsus (mm), and curved wing chord (mm). Of the 49 black skimmers examined, one adult and four hatch year birds were of unknown sex and were not included in this table.
| Sex/Age | Culmen | SD | Tarsus | SD | Wing Chord | SD |
| Female Adult (n = 3) | 63.30 | 10.08 | 33.60 | 2.96 | 367.00 | 22.91 |
| Female Hatch Year (n = 9) | 42.70 | 5.25 | 33.28 | 3.72 | 257.40 | 60.91 |
| Male Adult (n = 4) | 58.20 | 20.11 | 39.40 | 3.24 | 385.50 | 7.78 |
| Male Hatch Year (n = 28) | 47.40 | 8.74 | 35.68 | 3.79 | 270.80 | 74.73 |
Twenty-five of the 49 (51%) examined birds were in condition sufficient for organ assessment. The organs revealed various levels of atrophy, autolysis, and decomposition in most examined black skimmers. Specific organ findings were atrophied or dehydrated livers with sharp edges (40%, 10/25); pale/flaccid heart (32%, 8/25); abnormal cloacal or kidney region that appeared pale, autolyzed, or swollen (40%, 10/25); abnormal lungs that were autolyzed, contained fluid or plaques, or were hemorrhagic (36%, 9/25); or a combination of these descriptors (Table 3; Fig. 1).
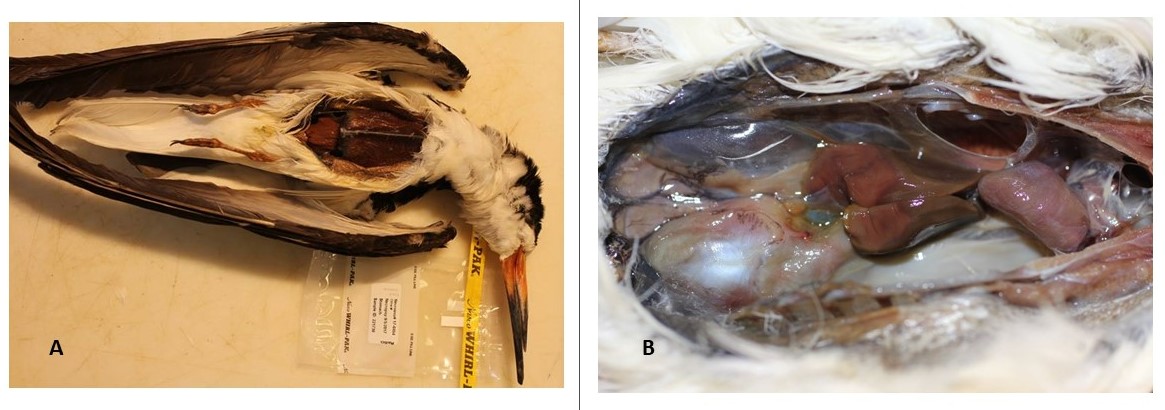
Table 3. Black skimmers (n = 25) age, sex, and inclusive body condition of individuals with external fouling, exhibited abnormal organs, and tested positive for gastrointestinal melena using occult blood test panels. 25/49 (51%) examined birds were in condition sufficient for organ assessment. (HY = hatch year, AD = adult, M = male, F = female)
| Age | Sex | Inclusive Body Condition | External Fouling | Dehydrated Liver | Flaccid/Pale Heart | Affected Cloaca/Kidney | Multiple Autolyzed Organs | GI Melena | Affected Lungs |
| HY | M | moderate | x* | x | |||||
| HY | M | moderate | x* | ||||||
| HY | M | moderate | x** | ||||||
| HY | M | moderate | x | x | x | x | x | ||
| HY | M | poor | x | x | x | x | x | ||
| HY | M | poor | x | ||||||
| HY | M | poor | x | x | x | ||||
| HY | M | poor | x | ||||||
| HY | M | poor | x | x | x | x | x | ||
| HY | M | poor | x | x | |||||
| HY | M | poor | x | ||||||
| HY | M | poor | x | ||||||
| HY | M | poor | x | ||||||
| HY | M | poor | x | x | |||||
| HY | M | poor | x | x | |||||
| HY | F | moderate | x | x | |||||
| HY | F | moderate | x | ||||||
| HY | F | poor | x | x | x | x | |||
| HY | F | poor | x | ||||||
| HY | F | poor | x | ||||||
| HY | F | poor | x | x | x | x | x | ||
| HY | U | poor | x | ||||||
| AD | M | moderate | x | ||||||
| AD | M | poor | x | x | x | x | x | ||
| AD | F | moderate | x | x |
** Indicates bird that has been euthanized.
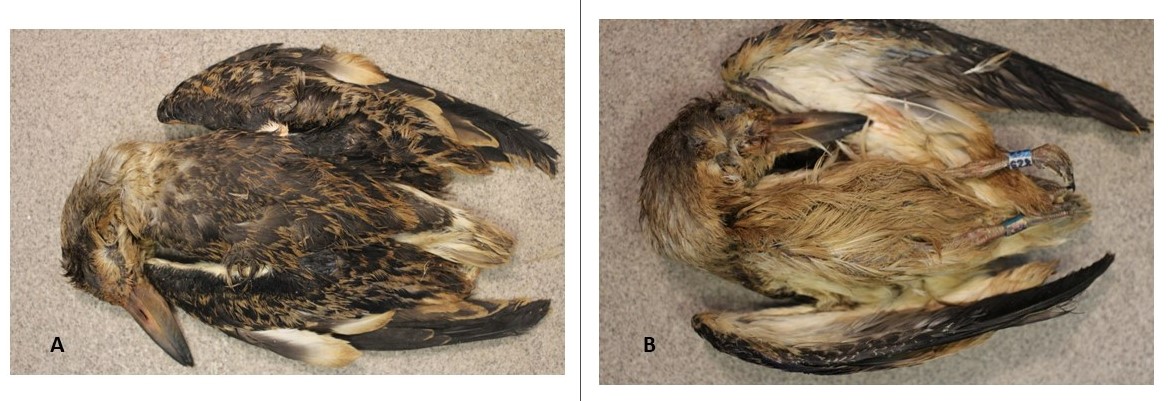
Six individuals (12%, 6/49) had evidence of external oil fouling (one adult, five hatch year; Table 3). Three of the six birds (50%) were externally fouled in small surface areas by crude oil and one (17%) had a greenish patch on the ventral feathers that appeared to be algal or biological fouling. Of the six fouled birds, two (33%) were covered in a substance that was yellowish and transparent in color but did not exhibit the physical characteristics of crude oil from DWH, noted as dark or reddish brown in color (Deepwater Horizon Natural Resource Damage Assessment Trustees 2016; Fig. 2).
Gastrointestinal Processing, Testing, and Sorting
GI examination revealed normal digesta in most individuals. However, seven carcasses (14%, 7/49) had dark and/or grainy substances in the intestinal region, and one carcass had this material in both stomach chambers. Based upon consistency and appearance, this material was determined to be melena, which was confirmed using occult tests in all cases (Table 3). Internal petroleum or synthetic odor were not found in the GI of any of the sampled birds. Additionally, no ingested oil, ulcers, erosions, or obvious lesions were reported in the examined GI tracts. The stomach contents included small, transparent fish bones, sand granules, nematodes, and ingested fish in various stages of digestion. Fifteen birds had empty stomach chambers, and no visible plastics or other petroleum products were found in any of the examined birds.
Discussion
Understanding the full impact of oil spills on wildlife is critical for the development of thorough damage assessments and restoration plans and helps inform future oil spill response and rehabilitation. Here, we performed necropsies on black skimmers recovered from the largest oil spill in the U.S., DWH, to assess internal injury, external fouling, and ingestion of plastics.
We report minimal evidence of external fouling of the black skimmers examined and that 41% (20/49) were hatch-year males in poor body condition (Tables 1, 3). To qualify for carcass collection from oil-impacted beaches, birds had to be in intact body condition for necropsy and tissue examination. Therefore, the number of examined birds (n = 49) and those in condition for organ assessment (25/49) are representative of a subsample of the total affected population that did not display acute lethal fouling of crude oil and could explain the lack of external fouling. In May 2011, the Natural Resource Damage Assessment (NRDA) reported deceased black skimmers (and many other seabird species) that were not visibly fouled by oil (USFWS 2011). Of the 253 (live and dead) black skimmers collected during DWH, 55 were visibly oiled, 153 were not visibly oiled, and 45 were of unknown oil status (internal examinations were not reported). The report cautioned that the 253 collected birds did not likely reflect the grand total number of black skimmers affected by DWH and the tallies were ongoing for years due to the nature of measuring spill impacts (USFWS 2011). Geographically, our samples may only be reflective of the black skimmers within the limited range of collection, even though this species inhabits a greater geographic span along the Gulf Coast.
Additional seabird mortality attributed to oil without evidence of external fouling was prevalent in DWH (45% of n = 6429; USFWS collected) where the frequency of deceased birds categorized as “dead and not visibly oiled” was greater than 1.5 times the number of birds found “dead and visibly oiled” (Tran et al. 2014). The same study analyzed USFWS collection data and suggested that while externally fouled birds are thought of as the most impacted during oil spills, perhaps the most pernicious impacts are invisible and internal (Tran et al. 2014). In a study that conducted external and blood evaluations on oil-exposed live birds during DWH, Fallon et al. (2017) reported hematologic injury in seabirds despite no visible oil. They concluded that even a small amount of external oil exposure was associated with hemolytic anemia in black skimmers and other species from DWH (Fallon et al. 2017). Hemolytic anemia has been associated with induced fatigue and reduced energy for efficient metabolic functioning of seabirds, potentially decreasing long-term survival (Butler et al. 1986). From the more recent Refugio oil spill, Fiorello et al. (2017) reported long-term impacts (significantly greater mean heterophil counts, heterophil to lymphocyte ratios, and total protein levels than the control group) in oil rehabilitated brown pelicans that were recaptured and evaluated post-release. In earlier spills, it has been estimated that 20% of bird mortality was due to oiling, in birds without evidence of external fouling. Specifically, Vauk et al. (1989) reported that while 80% of externally oiled birds contained oil in their digestive tract, 20% (179/365) of externally clean birds exhibited internal oiling. These findings suggest that external fouling alone is not indicative of the total number of affected individuals, whereas internal examination, blood evaluations and post-release evaluations likely provide a better proxy of the total number of individuals impacted by a spill.
While we did not find direct evidence of ingested petroleum oil or plastics, most of the birds were in poor body condition and internal organs were atrophied and dehydrated in appearance. Some of the carcasses were decomposed, and in various stages of autolysis. Decomposition and autolysis may have occurred prior to collection of carcasses, while dehydrated and atrophied organs may be a byproduct of long-term freezing. Alternatively, dehydration and atrophy of organs may be reflective of acute damage from oil exposure. Dehydration and emaciation, including loss of subcutaneous and abdominal fat, have been reported as the main gross lesions in alcids affected by oil spills in the past (Balseiro et al. 2005; Khan and Ryan 1991). Oil exposure in seabirds has also been found to promote poor body condition, which in turn, has been attributed to inefficient foraging effort and organ tissue damage, particularly of the alimentary tract and liver, as a result of PAH toxicity (Leighton 1993; Briggs et al. 1997). Toxicity from these persistent organic pollutants, such as PAHs continue to contaminate the liver months after the bird’s initial exposure (Troisi et al. 2006), potentially influencing recovery in oil exposed individuals, and contributing to lowered reproductive success of the population over time. Potentially, black skimmers with abnormal organs may be indicative of low-level exposure to oil through dermal (light feather fouling), respiratory, or oral contact (during foraging, preening, or from contaminated prey).
We did not detect visible plastic particles during gross necropsy; however, we cannot confirm the absence of microplastics small enough to remain undetected by the binocular scope. The smallest microplastic particles are theorized to be able to pass through tissues and cell membranes, but the impact on marine wildlife remains uncertain (Kühn and van Franeker 2020). Detection of these types of particles was beyond the scope of our study and not available for the majority of seabird ingestion research to date (Kühn and van Franeker 2020). The detection of microplastics and its associated impacts on marine organisms is an emerging field and a lack of published, standardized approaches to monitoring microplastic ingestion in seabirds remains (Vandermeersch et al. 2015; Wesch et al. 2016). For other marine biota, Wesch et al. (2016) conducted a literature review of monitoring ingestion of microplastics by marine biota and revealed that visual sorting was the most common identification method. Alternatively, Löder and Gerdts (2015) suggested that visual identification should not be solely relayed upon for microfiber particles that are <500 μm to avoid misidentification, however, this size threshold has not been substantiated by other studies. Microfibers are easily airborne, omnipresent in the environment, and bioavailable to marine organisms (Cole et al. 2011; Carney et al. 2018; Mishra et al. 2019); therefore, it is extremely difficult to distinguish between ingested fibers versus those that are present due to contamination of samples (Kühn and van Franeker 2020). Overestimation of these particles as ingestion items instead of secondary contamination of samples (e.g., from clothing, laboratory items) can occur without extensive laboratory controls (Wesch et al. 2016, Kühn and van Franeker 2020). It is possible that ingested microfibers were present in our black skimmer samples and went undetected (Wesch et al. 2016; Kühn and van Franeker 2020). However, if black skimmers foraging strategy potentially expose them to fibers and other microplastics at the water’s surface or through secondary ingestion of their fish prey that are too small for visible detection, further laboratory analyses and funds for testing are warranted. To accurately detect these particles, it has been suggested that spectroscopic analyses (e.g., Fourier transform infrared or Raman spectroscopy) should accompany visual identification and potential sources of contamination need to be accounted for and prevented to ensure accurate ingestion results (Wesch et al. 2016; Bradley et al. 2017; Michishita et al. 2023). As the status of ingested plastics for black skimmers may have changed over the past 11 years since the birds were collected and current examinations utilizing multiple detection techniques are needed to better account for the prevalence and potential impacts of microplastics on this species.
One black skimmer examined in our study had a substantial surface area of external fouling from a suspected synthetic compound. While the substance did not appear to be crude oil, it is possible it may have been a chemical dispersant used in the oil clean-up effort based on the material consistency, although we were unable to test the substance. Dispersants, including Corexit EC9500a and 9527, were used in great quantities during the clean-up of DWH (Wise and Wise 2011). While we were unable to confirm with certainty if the fouling was dispersant or another chemical solvent, previous research of dispersant 9527 indicated that the toxicity of oil-Corexit mixtures is of similar toxicity to that of oil alone (Peakall and Mackay 1987). Other dispersants (Finasol OSR-5 or OSR-12) have demonstrated negative thermoregulatory impacts on birds, perhaps due to birds preening the pollutants further into their plumage after feather fouling (Jenssen and Ekker 1990). More recently, Whitmer et al. (2018) reported negative effects on waterproofing and feather geometry on common murres (Uria aalge) exposed to Corexit 9500A. As dispersants are often used during oil clean up, the continued examination of the impacts of dispersants on seabirds is needed. This may be particularly important if dispersants contribute to a reduction in long-term survival of seabirds. We are not certain if what we reported is dispersant, however, further investigation of how dispersants impact the thermoregulatory functionality of feathers, and the overall survival of different seabird species, is needed due to their use in oil spill response. While dispersants are often considered a necessity in oil spill response, improved understanding of the negative impacts on wildlife can inform care for oil and dispersant effected seabirds during oil spill response and rehabilitation. This type of research has the potential to guide efforts for manufacturers of dispersants for development of wildlife safe oil spill management applications.
The collection and examination of seabirds affected by oil spills assists in natural resource damage assessments and allows for detailed examination and reporting of injuries, including petroleum and petroleum product ingestion. Documenting injuries from anthropogenic activities has the potential to inform best achievable care protocols for oiled seabirds, including black skimmers, in rehabilitation facilities. While DWH opportunistically provided black skimmer carcasses for examination, beach-cast black skimmer carcasses or those that do not survive wildlife rehabilitation could be potential sources for sampling in future research. Black skimmers in this study were examined approximately 7 years after DWH and were subjected to long-term freezing and thawing that may have interfered with the detection of ingested oil and abnormal organ condition. Fresh carcasses free from potential artifacts of freezing are optimal for examinations that are more conclusive and informative for future oil spill responses; therefore, examination of collected carcasses should be completed promptly during or immediately after oil spill events. To maximize the value of retained carcasses from an oil spill event, we recommend that government response agencies systematically retain carcasses collected from both impacted habitats and rehabilitation centers. In many regions, examinations of retained carcasses are postponed until after litigation due to their status as legal evidence. We propose that damage assessment teams support timely examination of retained evidentiary carcasses as part of the oil spill response process in order to incorporate demographic data into restoration actions and inform rehabilitation efforts. As seabird habitats continue to be impacted by anthropogenic activities, including oiling, dispersants used during oil spill response, and plastic debris, it is crucial to assess the multiple threats inflicted on these species to facilitate future protection and mitigation efforts.
Acknowledgments
We thank M. Ziccardi for organizing the acquisition of the bird carcasses for this research, for logistical planning, and for involvement in moving this work forward. We thank M. Miller, G. Frankfurter, K. Mills. A. Wells, and A. Reed for necropsy assistance and expertise. We thank M. Hester and T. Mena for the grant and administrative assistance and on-going support. Finally, we thank the responders from DWH response for collection of carcasses and staff and volunteers at the Theodore Wildlife Rehabilitation Center for the care and collection of birds during DWH.
This project was funded by the California Department of Fish and Wildlife Oil Spill Response Trust Fund through the Oiled Wildlife Care Network at the Wildlife Health Center, School of Veterinary Medicine, University of California, Davis, Research Agreement Number 201703366-04. All applicable ethical guidelines for the use of birds in research have been followed, including those presented in the Ornithological Council’s “Guidelines to the Use of Wild Birds in Research. Necropsy facilities were provided by CDFW-MWVCRC, we thank L. Henkel for logistical and facility support.
Literature Cited
- Balseiro, A., A. Espí, I. Márquez, V. Pérez, M. C. Ferreras, J. F. García Marín, and J. M. Prieto. 2005. Pathological features in marine birds affected by the Prestige’s oil spill in the north of Spain. Journal of Wildlife Diseases 41(2):371–378.
- Beyer, J., H. C. Trannum, T. Bakke, P. V. Hodson, and T. K. Collier. 2016. Environmental effects of the Deepwater Horizon oil spill: a review. Marine Pollution Bulletin 110(1):28–51.
- Bradley, M., S. Sukumaran, S. Lowry, and S. Woods. 2017. Tracking microplastics in the environment via FT-IR. Microscopy 32(8):17–23.
- Briggs, K. T., M. E. Gershwin, and D. W. Anderson. 1997. Consequences of petrochemical ingestion and stress on the avian immune system. ICES Journal of Marine Science 54:718–725.
- Broughton, J. M. 1994. Size of the bursa of Fabricius in relation to gonad size and age in Laysan and black-footed albatrosses. Condor 96:203–207.
- Bursian, S. J., C. R. Alexander, D. Cacela, F. L. Cunningham, K. M Dean, B. S. Dorr, C. K. Ellis, C. A. Godard-Codding, C. G. Guglielmo, K. C. Hanson-Dorr, K. E. Harr, K. A. Healy, M. J. Hooper, K. E. Horak, J. P. Isanhart, L. V. Kennedy, J. E. Link, I. Maggini, J. K. Moye, C. R. Perez, C. A. Pritsos, S. A. Shriner, K. A. Trust, and P. L. Tuttle. 2017. Overview of avian toxicity studies for the Deepwater Horizon natural resource damage assessment. Ecotoxicology and Environmental Safety 146: 4–10.
- Butler, R. G., D. B. Peakall, F. A. Leighton, J. Borthwick, and R. S. Harmon. 1986. Effects of crude oil exposure on standard metabolic rate of Leach’s storm-petrel. Condor 88:248–249.
- Carney Almroth, B. M., L. Åström, S. Roslund, H. Petersson, M. Johansson, and N. K. Persson. 2018. Quantifying shedding of synthetic fibers from textiles; a source of microplastics released into the environment. Environmental Science and Pollution Research 25:1191–1199.
- Cole, M., P. Lindeque, C. S. Halsband, and T. Galloway. 2011. Microplastics as contaminants in the marine environment: a review. Marine Pollution Bulletin 62(12):2588–2597.
- Deepwater Horizon Natural Resource Damage Assessment Trustees. 2016. Deepwater Horizon Oil Spill: Final Programmatic Damage Assessment and Restoration Plan and Final Programmatic Environmental Impact Statement. National Oceanic and Atmospheric Administration, Silver Spring, MD, USA. Available from: http://www.gulfspillrestoration.noaa.gov/restoration-planning/gulf-plan/
- Fallon, J. A., E. P. Smith, N. Schoch, J. D. Paruk, E. A. Adams, D. C. Evers, P. G. R. Jodice, C. Perkins, S. Schulte, and W. A. Hopkins. 2017. Hematological indices of injury to lightly oiled birds from the Deepwater Horizon oil spill. Environmental Toxicology and Chemistry 37(2):451–461.
- Fiorello, C., P. Jodice, J. Lamb, Y. Satge, K. Mills-Parker, D. Jaques, L. Henkel, R. Golightly, and M. Ziccardi. 2017. Post-release monitoring of oiled brown pelicans from the 2015 Refugio oil spill. International Oil Spill Conference Proceedings 2017(1):605–617. doi.org/10.7901/2169-3358-2017.1.605
- Furfey, B. C. 2014. The reproductive and foraging ecology of black skimmers (Rynchops niger) on a barrier island refuge in coastal Louisiana. Dissertation, Arkansas State University, Jonesboro, AR, USA
- Gilson, S. D., B. B. Parker, and D. C. Twedt. 1990. Evaluation of two commercial test kits for the detection of occult blood feces of dogs. American Journal of Veterinary Research 51(9):1385–1387.
- Hammer, S., R. G. Nager, P. C. D. Johnson, R. W. Furness, and J. F. Provencher. 2016. Plastic debris in great skua (Stercorarius skua) pellets corresponds to seabird prey species. Marine Pollution Bulletin 103:206–210.
- Jenssen, B. M., and M. Ekker. 1990. Effects of plumage contamination with crude oil dispersant mixtures on thermoregulation in common eiders and mallards. Archives of Environmental Contamination and Toxicology 20(3):398–403.
- Khan, R. A., and P. Ryan. 1991. Long term effects of crude oil on common murres (Uria aalge) following rehabilitation. Bulletin of Environmental Contamination and Toxicology 46:216–222.
- Kühn, S., and J. A. van Franeker. 2020. Quantitative overview of marine debris ingested by marine megafauna. Marine Pollution Bulletin 151:e110858.
- Leighton, F. A. 1993. The toxicity of petroleum oils to birds. Environmental Reviews 1(2):92–103.
- Löder M. G. J., and G. Gerdts. 2015. Methodology used for the detection and identification of microplastics—a critical appraisal. Pages 201–227 in M. Bergmann, L. Gutow, and M. Klages, editors. Marine Anthropogenic Litter. Springer, New York, NY, USA. doi.org/10.1007/978-3-319-16510-3_8
- Michishita, S. C. Gibble, C. Tubbs, R. Felton, J. Gjeltema, J. Lang, and M. Finkelstein. 2023. Microplastic in northern anchovies (Engraulis mordax) and common murres (Uria aalge) from the Monterey Bay, California USA: insights into prevalence, composition, and estrogenic activity. Environmental Pollution 316:120548.
- Mishra, S., C. C. Rath, and A. P. Das. 2019. Marine microfiber pollution: a review on present status and future challenges. Marine Pollution Bulletin 140:188–197.
- Paruk, J. D., E. M. Adams, H. Uher-Koch, K. A. Kovach, D. Long, C. Perkins, N. Schoch, and D. C. Evers. 2016. Polycyclic aromatic hydrocarbons in blood related to lower body mass in common loons. Science of the Total Environment 565:360–368.
- Peakall, D. B., and P. G. Mackay. 1987. A hazard assessment of chemically dispersed oil spills and seabirds. Marine Environmental Research 22(2):91–106.
- Pyle, P. 2009. Age determination and molt strategies in North American alcids. Marine Ornithology 37:219–225.
- Rapp, D. C., S. M Youngren, P. Hartzell, and K. D. Hyrenbach. 2017. Community-wide patterns of plastic ingestion in seabirds breeding at French Frigate Shoals, Northwestern Hawaiian Islands. Marine Pollution Bulletin 123:269–278.
- Savoca, M. S., M. E. Wohlfeil, S. E. Ebeler, and G. A. Nevitt. 2016. Marine plastic debris emits a keystone info-chemical for olfactory foraging seabirds. Science Advances 2:e1600395.
- Sileo, L., P. R. Sivert, and M. D. Samuel. 1989. Prevalence and characteristics of plastic ingested by Hawaiian seabirds. Pages 665–681 in R. S. Shomura and M. L. Godfrey, editors. Proceedings of the Second International Conference on Marine Debris, 2–7 April 1989, Honolulu, Hawaii. National Oceanic and Atmospheric Administration, Silver Spring, MD, USA.
- Spear, L. B., D. G. Ainley, and C. A. Ribic. 1995. Incidence of plastic in seabirds from the tropical Pacific, 1984–91: relation with distribution of species, sex, age, season, year, and body weight. Marine Environmental Research 40:123–146.
- Szaro, R. C., M. P. Dieter, G. H. Heinz, and J. F. Ferrell. 1978. Effects of chronic ingestion of south Louisiana crude oil on mallard ducklings. Environmental Research 17(3):426–436.
- Tanaka, K., J. van Franeker, D. Tomohiro, and H. Takada. 2019. Piece-by-piece analysis of additives and manufacturing byproducts in plastics ingested by seabirds: implications for risk and exposure to seabirds. Marine Pollution Bulletin 145:36–41.
- Tran, T., A. Yazdanparast, and E. A. Suess. 2014. Effect of oil spill on birds: a graphical assay of the Deepwater Horizon oil spill’s impact on birds. Computational Statistics 29:133–140.
- Troisi, G. M., S. Bexton, and I. Robinson. 2006. Polyaromatic hydrocarbon and PAH metabolite burdens in oiled common guillemots (Uria aalge) stranded on the East Coast of England 2001–02. Environmental Science & Technology 40(24):7938–7943.
- U.S. Fish and Wildlife Service (USFWS). 2011. Deepwater Horizon bird impact data from the DOI-ERDC NRDA Database, retrieved 12 May 2011. U.S. Geological Survey, Reston, VA, USA. Available from: https://www.fws.gov/sites/default/files/documents/Bird%20Data%20Species%20Spreadsheet%2005122011.pdf
- Valero, M. A., N. Gironès, M. A. García-Bodelón, M. V. Periago, I. Chico-Calero, M. Khoubbane, M. Fresno, and S. Mas-Coma. 2008. Anaemia in advanced chronic fasciolosis. Acta Tropica 108(1):35–43.
- Vandermeersch, G., L. Van Cauwenberghe, C. R. Janssen, A. Marques, K. Granby, G. Fait, M. J. J. Kotterman, J. Diogène, K. Bekaert, J. Robbens, and L. Devriese. 2015. A critical view on microplastic quantification in aquatic organisms. Environmental Research 143(B):46–55.
- van Franeker, J. A. 2004. Save the North Sea Fulmar-Litter-EcoQO Manual. Part 1: Collection and Dissection Procedures. Wageningen Environmental Research (Alterra), Wageningen, Netherlands.
- Vauk, G., E. Hartwig, B. Reineking, and E. Vauk-Hentzelt. 1989. Losses of seabirds by oil pollution at the German North Sea coast. Pages 749–754 in J. D. Ros, editor. Topics in Marine Biology: Proceedings of the 22nd European Marine Biology Symposium, Barcelona, Spain, August 1987. Scientia Marina, Barcelona, Spain.
- Wesch, C., K. Bredimus, M. Paulus, and R. Klein. 2016. Towards the suitable monitoring of ingestion of microplastics by marine biota: a review. Environmental Pollution 218:1200–1208.
- Whitmer, E. R., B. A. Elias, D. J. Harvey, and M. H. Ziccardi. 2018. An experimental study of the effects of chemically dispersed oil on feather structure and waterproofing in common murres (Uria aalge). Journal of Wildlife Diseases 54(2):315–328.
- Wilson, I. D. 1990. Hematemesis, melena, and hematochezia. Pages 439–442 in H. K. Walker, W. D. Hall, and J. W. Hurst, editors. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd edition. Butterworths, Boston, MA, USA.
- Wise, J., and J. P. Wise. 2011. A review of the toxicity of chemical dispersants. Reviews on Environmental Health 26(4):281–300.

