FULL RESEARCH ARTICLE
Alexis Leviner1 and John D. Perrine2*
1 California Polytechnic State University, Animal Sciences Department, 1 Grand Avenue, San Luis Obispo, CA 93407, USA
2 California Polytechnic State University, Biological Sciences Department, 1 Grand Avenue, San Luis Obispo, CA 93407, USA
*Corresponding Author: jperrine@calpoly.edu
Published 10 July 2023 • doi.org/10.51492/cfwj.109.6
Abstract
Microplastics are persistent environmental contaminants that to date have been studied primarily in aquatic systems, but few studies have examined their prevalence or impact in terrestrial trophic networks. A recent study documented microplastics in the gastrointestinal (GI) tracts of terrestrial raptors in central Florida. We used protocols based on that study to develop a baseline dataset of microplastics in the GI tracts of terrestrial raptors on the central coast of California. In 2021, Pacific Wildlife Care, a wildlife rehabilitation center in Morro Bay, CA, provided 16 raptor carcasses: three red-tailed hawks (Buteo jamaicensis), four red-shouldered hawks (Buteo lineatus), two great horned owls (Bubo virginianus), and seven barn owls (Tyto alba). We found microplastics in the GI tracts of all 16 birds, with a mean (± SE) of 12.25 (± 1.89) microplastic particles per bird. Of the 196 microplastic particles we observed, microfibers were the most abundant (58%), followed by microbeads (34%), and microfragments (8%). However, microbeads were the most prevalent, found in all 16 birds, followed by microfibers (15 birds), and microfragments (6 birds). The chemical composition and source of these contaminants remains unclear, along with the physiological and ecological implications to raptor populations and their ecosystems, and the extent to which microplastics occur in other terrestrial species and regions in California.
Key Words: birds, conservation, ecology, ecotoxicology, hawks, ingestion, owls, pollution
| Citation: A. Leviner, and J. D. Perrine. 2023. Documentation of microplastics in the gastrointestinal tracts of terrestrial raptors in central California, USA. California Fish and Wildlife Journal 109:e6. |
| Editor: Shannon Skalos, Wildlife Branch |
| Submitted: 10 March 2023; Accepted: 19 April 2023 |
| Copyright: ©2023, Leviner and Perrine. This is an open access article and is considered public domain. Users have the right to read, download, copy, distribute, print, search, or link to the full texts of articles in this journal, crawl them for indexing, pass them as data to software, or use them for any other lawful purpose, provided the authors and the California Department of Fish and Wildlife are acknowledged. |
| Funding: This project was funded primarily by the William and Linda Frost Summer Scholars Program at the College of Science and Mathematics at California Polytechnic State University, San Luis Obispo. |
| Competing Interests: The authors have not declared any competing interests. |
Introduction
Microplastics (MPs), defined as small plastic particles < 5 mm and > 0.1 nm, have been found in terrestrial and aquatic environments worldwide and are believed to be accumulating at an accelerating rate (Thompson 2015; Wong et al. 2020; Prata et al. 2021). Microplastics may occur as a primary source from the production of small-sized particles, or as a secondary source via degradation or fragmentation of larger plastics (Casillas et al. 2023). Collectively, MPs are a highly diverse form of contaminants with complex composition and take many shapes such as spheres, fragments, and fibers (Vethaak and Legler 2021). Their persistence in the environment results in the “plastic cycle”, the movement of MPs between the air, the ocean, freshwater bodies, sediments, and terrestrial habitats (Bank and Hansson 2019), and leads to inevitable exposure to humans and other biota (Prata et al. 2021).
Microplastics have been documented to cause a wide range of toxic and physiological effects when ingested by animals, including reduced immune function, increased inflammatory response, organ failure, and endocrine disruption (Teuten et al. 2009; Prata et al. 2021; Wang et al. 2021; de Souza et al. 2022). Plastics can also contain additional toxicants such as phthalates, stabilizers, fire retardants, and dyes added during manufacturing (Teuten et al. 2009; Wang et al. 2021). Furthermore, MPs can absorb and concentrate other potential toxicants from the environment (Prokić et al. 2019; Xu et al. 2020; Prata et al. 2021), resulting in multiple and potentially synergistic toxic effects when ingested. However, much of the MP research to date has focused on marine and aquatic systems. For example, in a recent review, only 13% (65/500) of analyzed studies dealt with the exposure or effects of MPs on terrestrial species, with almost all of these performed on soil invertebrates (Prokić et al. 2021). For birds, this trend is even more pronounced, with the vast majority of studies of MP exposure conducted on marine and aquatic birds (Wang et al. 2021). Given their long lives, long-distance movements, and importance as a top predator in many ecosystems, raptors in particular warrant further study (Carlin et al. 2020; Wang et al. 2021).
Recently, Carlin et al. (2020) documented MPs in the gastrointestinal (GI) tracts of several raptor species in central Florida, one of the first studies to assess MPs in North American terrestrial birds. To date, no published study has examined MPs in California raptors. Our objective was to establish a baseline data set documenting MPs in the GI tracts of terrestrial raptors on the central coast of California.
Methods
Specimen Collection
Specimen collection, post-mortem sampling, and chemical digestion methods were based on those used by Carlin et al. (2020). We examined birds provided by Pacific Wildlife Care (PWC), a wildlife rehabilitation center in Morro Bay, CA, USA, under the strict requirement that they had not consumed any human-provisioned feed while in captivity. PWC staff collected these birds between January and June 2021 from various locations on California’s central coast (35.303, –120.660), ranging from San Simeon to Nipomo (Table 1). PWC staff collected one specimen (CP-958) dead following a window strike; the others were collected alive but died shortly after collection (CP-942) or were euthanized within 24 hours of intake due to poor prospects for recovery. No birds were sacrificed specifically for this study. The carcasses were frozen immediately after death to minimize decomposition. PWC provided records of each bird’s collection location, dates of collection and death, age class, body mass and condition, and associated pathologies and diagnoses.
Table 1. Characteristics of raptor specimens from California’s central coast, with quantity and type of microplastics found in their gastrointestinal tracts, February ̶ August 2021. BANO = barn owl, GHOW = great horned owl, RSHA = red-shouldered hawk, RTHA = red-tailed hawk. “Locality” indicates the municipality where the specimen was collected. “PWC-ID” and “CP-ID” are case and specimen tracking numbers for Pacific Wildlife Care and the California State Polytechnic University, San Luis Obispo Vertebrate Collection, respectively.
| Species | Age Class | Sex | Locality | Date Collected | PWC-ID | CP-ID | Fibers | Beads | Fragments | Total MPs |
| BANO | Adult | ? | Nipomo | 14 Jan 2021 | 21-64 | 937 | 14 | 5 | 1 | 20 |
| BANO | Adult | M | Santa Margarita | 30 Jan 2021 | 21-124 | 943 | 11 | 1 | 0 | 12 |
| BANO | Adult | M | Templeton | 24 Feb 2021 | 21-189 | 954 | 4 | 2 | 0 | 6 |
| BANO | Adult | M | San Luis Obispo | 4 Mar 2021 | 21-211 | 956 | 8 | 8 | 0 | 16 |
| BANO | Adult | ? | San Simeon | 6 Mar 2021 | 21-217 | 955 | 3 | 1 | 0 | 4 |
| BANO | Adult | ? | San Simeon | 23 Mar 2021 | 21-306 | 953 | 1 | 2 | 0 | 3 |
| BANO | Adult | F | San Luis Obispo | 25 Mar 2021 | 21-313 | 952 | 0 | 4 | 1 | 5 |
| GHOW | Adult | ? | Paso Robles | 24 Jan 2021 | 21-102 | 941 | 15 | 2 | 2 | 19 |
| GHOW | Adult | ? | Paso Robles | 28 Jan 2021 | 21-114 | 942 | 10 | 8 | 0 | 18 |
| RSHA | Adult | ? | Oceano | 12 Feb 2021 | 21-164 | 960 | 16 | 8 | 7 | 31 |
| RSHA | Adult | M | Morro Bay | 2 Mar 2021 | 21-210 | 958 | 9 | 3 | 3 | 15 |
| RSHA | Adult | M | Pismo Beach | 2 Mar 2021 | 21-209 | 959 | 5 | 8 | 0 | 13 |
| RSHA | Adult | ? | Grover Beach | 6 Jun 2021 | 21-1097 | 967 | 4 | 5 | 0 | 9 |
| RTHA | Juvenile | M | Atascadero | 17 Jan 2021 | 21-79 | 939 | 5 | 2 | 1 | 8 |
| RTHA | Juvenile | M | Paso Robles | 24 Jan 2021 | 21-101 | 940 | 3 | 1 | 0 | 4 |
| RTHA | Fledgling | ? | Paso Robles | 13 Jun 2021 | 21-1164 | 970 | 6 | 7 | 0 | 13 |
The frozen carcasses were transported to the Vertebrate Zoology Lab at California Polytechnic State University, San Luis Obispo, CA, between February and June 2021. We conducted lab work between February and August 2021. We thawed each bird overnight and then extracted the complete GI tract, from the cranial aspect of the esophagus to the caudal connection of the large intestine to the cloaca. The GI tract was removed in one intact piece and was handled only with metal tools (never with a gloved hand) to prevent additional external MP contamination. Each GI tract was individually labeled, wrapped in aluminum foil, and then frozen until chemical digestion took place.
Chemical Digestion and Documentation of MPs
Digestion with potassium hydroxide (KOH) has been recommended as the standard method for extracting MPs from tissues because many plastics are resistant to it (Kühn et al. 2017; Thiele et al. 2019). To verify the efficacy of the protocols, we first conducted a pilot study in April ̶ May 2021 of five birds: one red-tailed hawk (Buteo jamaicensis), two great horned owls (Bubo virginianus), and two barn owls (Tyto alba); two of these birds were later discarded (see below). Once the protocols were verified, we examined an additional 13 birds in June ̶ August 2021: two red-tailed hawks, four red-shouldered hawks (Buteo lineatus), one great horned owl, and six barn owls, yielding a total of 16 usable specimens.
After we thawed each GI tract, we rinsed its outer surface with nanopure water and then placed it into a 500 mL Erlenmeyer flask along with a 10% KOH solution in a 3:1 ratio to the GI tract’s mass. We incubated the samples in a fume hood for 48 hours in a shaking incubator at 65 rpm and 40˚C, then removed them and allowed them to cool at room temperature for 24 hours. To separate the MPs from the digested tissues, each sample was vacuum filtered through 90 mm, 2.5 µm glass fiber filter paper, and the filtrate solution was then filtered a second time through 90 mm, 0.7 µm glass fiber filter paper. Following filtration, we immediately placed each filter paper into a triple-rinsed glass petri dish and allowed it to dry for 24 hours. We then examined each filter paper under a dissecting scope to quantify and describe MPs present. We examined each filter paper three times at 20x magnification to confirm that all MPs were accounted for, and total MP count was consistent. A single investigator (AL) conducted all examinations to prevent between-investigator biases and inconsistencies. We determined the quantity, color, and type of each MP following the guidelines of Sartain et al. (2018), and then photographed each MP at 30x magnification via the dissecting scope (Fig. 1). Lastly, we individually wrapped each filter paper in aluminum foil and stored them at room temperature for potential future identification of the MPs via micro-Fourier spectroscopy.
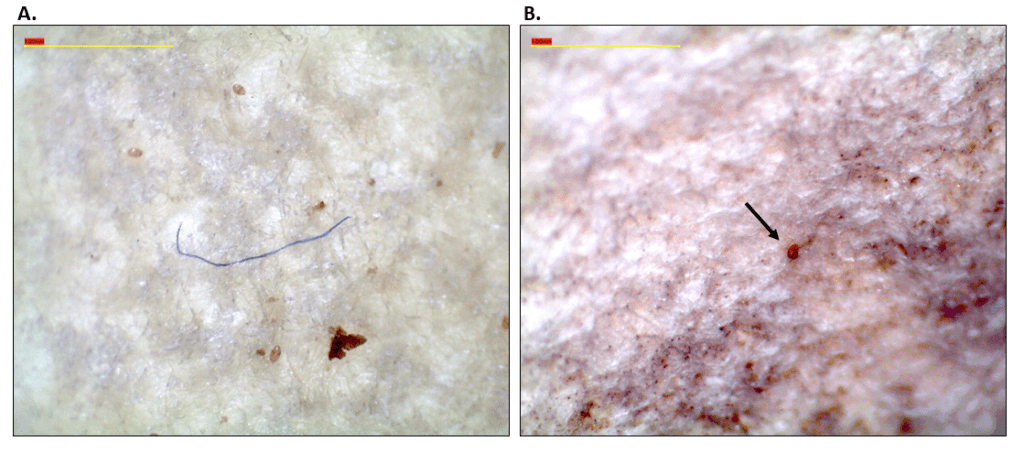
Positive and Contamination Controls
We used positive controls to verify that the digestion process successfully recovered MPs from tissues, and contamination controls to ensure that MPs from the surrounding environment were not contaminating our samples during processing. For the positive controls, we assessed two different protocols. First, we added eight polyester pieces to the GI tracts of a barn owl and a great horned owl prior to chemical digestion. However, we then learned that KOH could digest polyester, and we were concerned about differentiating our control MPs from MPs actually in the birds’ GI tracts, so we discontinued this approach and did not use these birds in our final data set. Our second approach was based on that used by Carlin et al. (2020): we placed 10 black polyethylene fibers < 3 mm in length into each of three separate 500 mL Erlenmeyer flasks with approximately 25 mL of 10% KOH and nanopure water solution. Following the standard chemical digestion protocol and filtration, we counted the number of fibers observed to estimate the loss that may occur during the chemical digestion procedure.
Our contamination controls were also based on those used by Carlin et al. (2020). For the first five birds we examined, we placed four filter papers around the dissecting scope, one per corner, for the entire duration of MP quantification. We then examined these filter papers for MPs in the same manner as for the filters from the birds’ GI tracts.
Results
In the three positive control trials, we recovered 9 of 10 polyethylene fibers from one sample, and 8 of 10 fibers from the other two samples, yielding an overall recovery rate of 83%. In our contamination control trials, the 20 contamination filters yielded an average of 0.3 (± 0.1 SE) MPs per filter. Filters with contaminants contained fibers or blue fragments which typically appeared thicker and more vibrantly colored than the fibers and fragments we found in the chemically-digested GI tracts of the birds. Based on the low amount of environmental contamination detected in our pilot project, we did not use contamination controls in the remaining 13 specimens.
We found MPs in the GI tracts of all 16 birds examined (Table 1), with a median of 12.5 and a mean (± SE) of 12.25 (± 1.89) particles per bird (Fig. 2). Of the total 196 MP particles observed, microfibers were the most abundant (58%), followed by microbeads (34%), and microfragments (8%). However, microbeads were the most prevalent, found in all 16 birds, followed by microfibers (in 15 birds), and microfragments (in 6 birds).
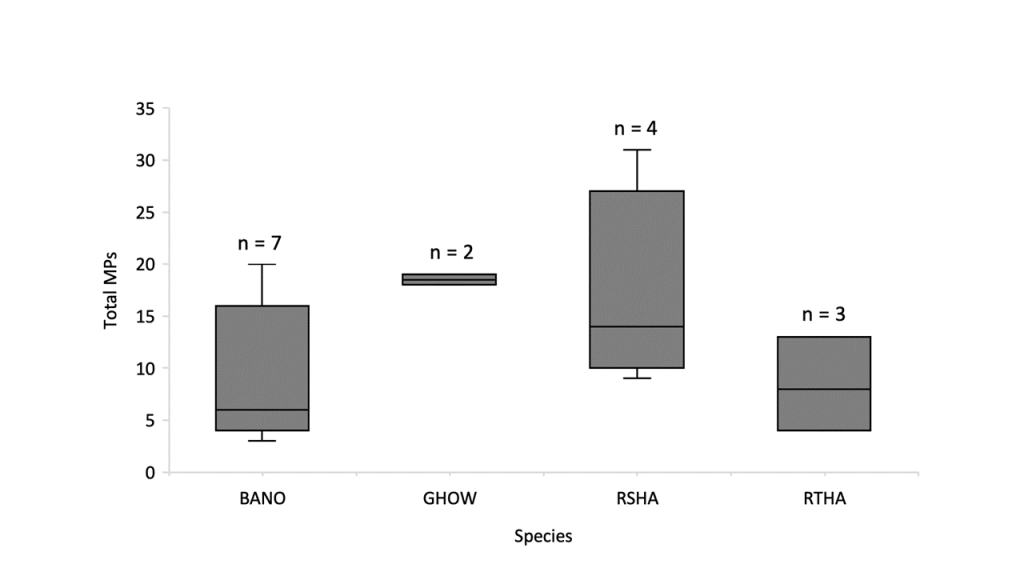
Discussion
All 16 raptors that we examined from California’s central coast contained MPs in their GI tracts. To our knowledge, this is the first published study of MPs in terrestrial raptors in California and adds to a small but growing literature documenting MPs in birds outside of marine or aquatic ecosystems. Zhao et al. (2016) found MPs in 16 of 17 birds of 12 different species, including raptors, wading birds, and passerines, collected in Shanghai, China. Hoang and Mitten (2022) found MPs in the GI tracts of all 14 individuals of 5 species of migratory passerines collected in Chicago, USA. Most similar to our study, Carlin et al. (2020) found MPs in the GI tracts of all 63 individuals of 8 predatory bird species, including hawks, owls, and vultures, collected in central Florida, USA. Due to the opportunistic nature of our study, our sample sizes were too small to assess any potential differences in MP quantity or types among species, sexes, age classes, or specific geographic locations (Prokić et al. 2021), but such differences would be straightforward to assess in the future if sufficient specimens are acquired. Even so, our study provides an important baseline of MP incidence and abundance in these taxa and adds to the growing number of studies indicating that MPs appear to be ubiquitous in many ecosystems, not just aquatic ones.
A thorough understanding of the prevalence of MPs in wild birds throughout California will require examining thousands of specimens. Wildlife rescue and rehabilitation facilities such as Pacific Wildlife Care can play a vital role in this effort, as statewide they handle tens of thousands of individuals of dozens of wild bird species every year (R. Duerr, International Bird Rescue, pers. comm.). If even a small proportion of the birds that died in captivity or were euthanized were made available for studies such as ours, this would constitute an invaluable source of specimens, and entail no additional mortality to wild populations. In addition, freshly killed wild birds could be salvaged from roadkills and window strikes (e.g., Hoang and Mitten 2022), or provided by sport hunters (especially waterfowl, gallinaceous and columbiform birds); however, examination of MPs in these specimens may be complicated by significant bodily damage not occurring in birds collected alive by wildlife rescue facilities. Regardless of the source of the specimens, research on wild birds will be an essential complement to further experimental studies of MP effects upon captive birds (e.g., de Sousa et al. 2021).
We have not yet determined the chemical composition of the MPs found in our sample birds, which could provide important insight into their source, mode of ingestion, and potential toxicity; such analysis could also indicate whether the MPs in our contamination controls were chemically different than those found in the birds’ GI tracts. Using micro-Fourier infrared spectroscopy, Carlin et al. (2020) found that most of the identifiable MPs in the GI tracts of predatory birds were composed of cellulose, polyethylene terephthalate (PET), or polymer blends. Our study is consistent with prior studies finding fibers to be prevalent in the GI tracts of terrestrial birds (Zhao et al. 2016; Carlin et al. 2020; Weitzel et al. 2021; Hoang and Mitten 2022). These fibers likely originate from washing clothes, with the fibers entering the aquatic environment via the effluent discharge from wastewater treatment facilities (Browne et al. 2011; McCormick et al. 2014). Unlike prior studies, microbeads were the most prevalent MP form in our birds, occurring in every specimen but typically in lower abundance than fibers. Although California and federal law have banned microbeads from personal cosmetic products such as facial cleansers and toothpaste, these bans do not apply to other sources such as deodorants, non-cosmetic cleaning products and industrial applications (Kettenmann 2016). Microbeads likely enter the ecosystem primarily via municipal wastewater discharge or the use of sewage sludge as agricultural fertilizer (An et al. 2020), but the extent to which they have pervaded terrestrial trophic networks is currently unknown.
Microplastics are likely ubiquitous in California’s estuaries and beaches (Sutton et al. 2016; Horn et al. 2019) but riparian and lacustrine systems in California have not yet been extensively sampled. Despite their proximity to the coast, the hawks and owls in our study primarily consume small terrestrial vertebrates such as rodents, birds, and snakes, so it remains unclear how they ingested MPs. Possible routes of MP ingestion by raptors include direct ingestion while drinking, secondary consumption of MPs within or upon prey species, or via some other mechanism such as grooming or acquiring nest materials. Although most studies to date have addressed MPs in aquatic environments, they may also be present in soils due to agricultural practices, pollution, and other mechanisms, with earthworms likely providing the entry point to the terrestrial food chain (Xu et al. 2020). For example, Carlin et al. (2020) found significantly more MPs in the terrestrial-feeding red-shouldered hawk than in the primarily piscivorous osprey (Pandion haliaetus). Trophic transfer of MPs, both within and across ecosystems, remains an important subject for further study (Nelms et al. 2018; Xu et al. 2020).
The physical, physiological, and ecological effects of MP ingestion by birds also remain unclear. Unlike the macroplastics (> 5 mm) ingested by seabirds (e.g., Fry et al. 1987; Pierce et al. 2004) and vultures (e.g., Houston et al. 2007), MPs found in the GI tracts of terrestrial birds seem to occur in a few tens of particles per individual, too few to cause mortality due to intestinal blockage or severely impacted digestion. However, ingestion of MPs can cause lesions, ulcers, and inflammation in birds’ GI tracts, along with physiological effects such as endocrine disruption, ultimately resulting in reduced growth, survival, and reproductive rates (Wang et al., 2021). Experimental ingestion of MPs by captive Japanese quail (Coturnix japonica) led to reduced body mass and altered physiological functions in the brain, liver, intestine, and gizzard (de Sousa et al. 2021). The magnitude of these effects likely depends on how long the MPs are retained in the bird’s GI tract, which is poorly known for most species (Provencher et al. 2017). Further study is needed to determine whether MP load correlates with reduced fitness metrics in wild terrestrial birds such as body size, gonad size or performance, fecundity, or longevity; what effect these changes might have upon bird populations; and the cascading effects within their ecological communities (Wang et al. 2021).
Worldwide production of plastics is continuing to increase exponentially, to an estimated 33 billion metric tons per year by 2050 (Miller et al. 2017). The abundance of MPs in the environment and their inevitable ingestion by wild species can also be expected to increase in coming years. For example, Wilcox et al. (2015) predict that 95% of individual birds of 99% of seabird species worldwide will have ingested plastic by 2050. A thorough understanding of the physiological and ecological impacts of MP consumption by wildlife is therefore essential and cannot be limited merely to aquatic filter-feeders and detritivores. As top avian predators that typically consume whole prey, raptors can be a valuable indicator of MP presence in terrestrial trophic networks. Our study provides a baseline of MP prevalence and abundance in terrestrial raptors on California’s central coast. Continued monitoring of raptors and other taxa throughout the state, in both terrestrial and aquatic environments, is warranted to guide and assess policies to manage the effects of plastic pollution.
Acknowledgments
This project was funded primarily by the William and Linda Frost Summer Scholars Program at the College of Science and Mathematics at California Polytechnic State University, San Luis Obispo. Special thanks to the staff at Pacific Wildlife Care, especially K. Boland, for providing bird carcasses for this study. A. Hamrick provided valuable assistance with laboratory logistics. S. Skalos and two anonymous reviewers provided feedback which significantly improved this manuscript.
Literature Cited
- An, L., Q. Liu, Y. Deng, W. Wu, Y. Gao, and W. Ling. 2020. Sources of microplastic in the environment. Pages 143–160 in D. He and Y. Luo, editors. Microplastics in Terrestrial Environments: Emerging Contaminants and Major Challenges. Handbook of Environmental Chemistry 95. Springer Nature, Cham, Switzerland.
- Bank, M. S., and S. V. Hansson. 2019. The plastic cycle: a novel and holistic paradigm for the Anthropocene. Environmental Science and Technology 53(13):7177–7179. doi.org/10.1021/acs.est.9b02942
- Browne, M. A., P. Crump, S. J. Niven, E. Teuten, A. Tonkin, T. Galloway, and R. Thompson. 2011. Accumulation of microplastic on shorelines worldwide: sources and sinks. Environmental Science and Technology 45(21):9175–9179. doi.org/10.1021/es201811s
- Carlin, J., C. Craig, S. Little, M. Donnelly, D. Fox, L. Zhai, and L. Walters. 2020. Microplastic accumulation in the gastrointestinal tracts in birds of prey in central Florida, USA. Environmental Pollution 264:114633. doi.org/10.1016/j.envpol.2020.114633
- Casillas, G., B. C. Hubbard, J. Telfer, M. Zarate-Bermudez, C. Muianga, G. M. Zarus, Y. Carroll, A. Ellis, and C. M. Hunter. 2023. Microplastics scoping review of environmental and human exposure data. Microplastics 2(1):78–92. doi.org/10.3390/microplastics2010006
- de Souza, S. S., Í. N. Freitas, S. de Oliveira Gonçalves, T. M. da Luz, A. P. da Costa Araújo, R. Rajagopal, G. Balasubramani, M. M. Rahman, and G. Malafaia. 2022. Toxicity induced via ingestion of naturally-aged polystyrene microplastics by a small-sized terrestrial bird and its potential role as vectors for the dispersion of these pollutants. Journal of Hazardous Materials 434:128814. doi.org/10.1016/j.jhazmat.2022.128814
- Fry, D. M., S. I. Fefer, and L. Sileo. 1987. Ingestion of plastic debris by Laysan albatrosses and wedge-tailed shearwaters in the Hawaiian Islands. Marine Pollution Bulletin 18(6):339–343. doi.org/10.1016/S0025-326X(87)80022-X
- Hoang, T., and S. Mitten. 2022. Microplastic accumulation in the gastrointestinal tracts of nestling and adult migratory birds. Science of the Total Environment 838:155827. doi.org/10.1016/j.scitotenv.2022.155827
- Horn, D., M. Miller, S. Anderson, and C. Steele. 2019. Microplastics are ubiquitous on California beaches and enter the coastal food web through consumption by Pacific mole crabs. Marine Pollution Bulletin 139:231–237. doi.org/10.1016/j.marpolbul.2018.12.039
- Houston, D. C., A. Mee, and M. McGrady. 2007. Why do condors and vultures eat junk? The implications for conservation. Journal of Raptor Research 41:235–238. doi.org/10.3356/0892-1016(2007)41[235:WDCAVE]2.0.CO;2
- Kettenmann, S. 2016. Nationwide ban on plastic microbeads in cosmetics. Natural Resources and Environment 31:58–59.
- Kühn, S., B. van Werven, A. van Oyen, A. Meijboom, E. L Bravo Rebolledo, and J. A. van Franeker. 2017. The use of potassium hydroxide (KOH) solution as a suitable approach to isolate plastics ingested by marine organisms. Marine Pollution Bulletin 115:86–90. doi.org/10.1016/j.marpolbul.2016.11.034
- McCormick, A., T. J. Hoellein, S. A. Mason, J. Schluep, and J. J. Kelly. 2014. Microplastic is an abundant and distinct microbial habitat in an urban river. Environmental Science and Technology 48:11863–11871. doi.org/10.1021/es503610r
- Miller, M. E., F. J. Kroon, and C. A. Motti. 2017. Recovering microplastics from marine samples: a review of current practices. Marine Pollution Bulletin 123:6–18. doi.org/10.1016/j.marpolbul.2017.08.058
- Nelms, S. E., T. S. Galloway, B. J. Godley, D. S. Jarvis, and P. K. Lindeque. 2018. Investigating microplastic trophic transfer in marine top predators. Environmental Pollution 238:999–1007. doi.org/10.1016/j.envpol.2018.02.016
- Pierce, K. E., R. J. Harris, L. S. Larned, and M. A. Pokras. 2004. Obstruction and starvation associated with plastic ingestion in a northern gannet Morus bassanus and a greater shearwater Puffinus gravis. Marine Ornithology 32:187–189.
- Prata, J. C., J. P. da Costa, I. Lopes, A. L. Andrady, A. C. Duarte, and T. Rocha-Santos. 2021. A One Health perspective of the impacts of microplastics on animal, human and environmental health. Science of the Total Environment 777:146094. doi.org/10.1016/j.scitotenv.2021.146094
- Prokić, M. D., B. R. Gavrilović, T. B. Radovanović, J. P. Gavrić, T. G. Petrović, S. G. Despotović, and C. Faggio. 2021. Studying microplastics: Lessons from evaluated literature on animal model organisms and experimental approaches. Journal of Hazardous Materials 414:125476. doi.org/10.1016/j.jhazmat.2021.125476
- Prokić, M. D., T. B. Radovanović, J. P. Gavrić, and C. Faggio. 2019. Ecotoxicological effects of microplastics: examination of biomarkers, current state and future perspectives. Trends in Analytical Chemistry 111:37–46. doi.org/10.1016/j.trac.2018.12.001
- Provencher, J. F., A. L. Bond, S. Avery-Gomm, S. B. Borrelle, E. L. Bravo Rebolledo, S. Hammer, S. Kühn, J. L. Lavers, M. L. Mallory, A. Trevail, and J. A. van Franeker. 2017. Quantifying ingested debris in marine megafauna: a review and recommendations for standardization. Analytical Methods 9:1454–1469. doi.org/10.1039/c6ay02419j
- Sutton, R., S. A. Mason, S. K. Stanek, E. Willis-Norton, I. F. Wren, and C. Box. 2016. Microplastic contamination in the San Francisco Bay, California, USA. Marine Pollution Bulletin 109:230–235. doi.org/10.1016/j.marpolbul.2016.05.077
- Teuten, E. L., J. M. Saquing, D. R. U. Knappe, M. A. Barlaz, S. Jonsson, A. Björn, S. J. Rowland, R. C. Thompson, T. S. Galloway, R. Yamashita, D. Ochi, Y. Watanuki, C. Moore, P. H. Viet, T. S. Tana, M. Prudente, R. Boonyatumanond, M. P. Zakaria, K. Akkhavong, Y. Ogata, H. Hirai, S. Iwasa, K. Mizukawa, Y. Hagino, A. Imamura, M. Saha, and H. Takada. 2009. Transport and release of chemicals from plastics to the environment and to wildlife. Philosophical Transactions of the Royal Society B 364:2027–2045. doi.org/10.1098/rstb.2008.0284
- Thiele, C. J., M. D. Hudson, and A. E. Russell. 2019. Evaluation of existing methods to extract microplastics from bivalve tissue: adapted KOH digestion protocol improves filtration at single-digit pore size. Marine Pollution Bulletin 142:384–393. doi.org/10.1016/j.marpolbul.2019.03.003
- Thompson, R. C. 2015. Microplastics in the marine environment: sources, consequences and solutions. Pages 185–200 in M. Bergmann, L. Gutow, and M. Klages, editors. Marine Anthropogenic Litter. Springer, New York, NY, USA. doi.org/10.1007/978-3-319-16510-3
- Vethaak, A. D. and J. Legler. 2021. Microplastics and human health. Science 371:672–674. doi.org/10.1126/science.abe5041
- Wang, L., G. Nabi, L. Yin, Y. Wang, S. Li, Z. Hao, and D. Li. 2021. Birds and plastic pollution: recent advances. Avian Research 12:1–9. doi.org/10.1186/s40657-021-00293-2
- Weitzel, S. L., J. M. Feura, S. A. Rush, R. B. Iglay, and M. S. Woodrey. 2021. Availability and assessment of microplastic ingestion by marsh birds in Mississippi Gulf Coast tidal marshes. Marine Pollution Bulletin 166:112187. doi.org/10.1016/j.marpolbul.2021.112187
- Wilcox, C., E. van Sebille, and B. D. Hardesty. 2015. Threat of plastic pollution to seabirds is global, pervasive, and increasing. Proceedings of the National Academy of Sciences 112:11899–11904. doi.org/10.1073/pnas.1502108112
- Wong, S. L., B. B. Nyakuma, K. Y. Wong, C. T. Lee, T. H. Lee, and C. H. Lee. 2020. Microplastics and nanoplastics in global food webs: a bibliometric analysis (2009–2019). Marine Pollution Bulletin 158:111432. doi.org/10.1016/j.marpolbul.2020.111432
- Xu, B., F. Liu, Z. Cryder, D. Huang, Z. Lu, Y. He, H. Wang, Z. Lu, P. C. Brookes, C. Tang, J. Gan and J. Xu. 2020. Microplastics in the soil environment: occurrence, risks, interactions and fate—a review. Critical Reviews in Environmental Science and Technology 50:2175–2222. doi.org/10.1080/10643389.2019.1694822
- Zhao, S., L. Zhu, and D. Li. 2016. Microscopic anthropogenic litter in terrestrial birds from Shanghai, China: not only plastics but also natural fibers. Science of the Total Environment 550:1110–1115. doi.org/10.1016/j.scitotenv.2016.01.112

