FULL RESEARCH ARTICLE
John Scott Foott1*, Jason Kindopp2, Katie Gordon2,3, Alana Imrie2,3, and Kassie Hikey2
1 U.S. Fish and Wildlife Service, Pacific Southwest Region, 24411 Coleman Hatchery Road, Anderson, CA 96007, USA (Retired)
2 California Department of Water Resources, 460 Glen Drive, Oroville, CA 95966, USA
3 Pacific States Marine Fisheries Commission, 205 SE Spokane Street, Suite 100, Portland, OR 97202, USA
*Corresponding Author: jsfoott@gmail.com
Published 10 July 2023 • doi.org/10.51492/cfwj.109.9
Abstract
We performed a five-year (2015–2020) survey of juvenile natural Chinook Salmon (Oncorhynchus tshawytscha), adult salmon carcasses, and river water from the lower Feather River to determine infection prevalence, distribution, and spore quantity of the myxozoan parasite Ceratonova shasta. Average prevalence of infection in juvenile salmon collected from the high flow channel ranged from 45–58% depending on assay method. Initial infection of fry and detection of actinospore stage in river water began in late January or early February. Overt disease occurred in March and was lethal. Infection of the gill was detected weeks ahead of intestinal infection. Water-borne spore measurement and fish infection demonstrate an infectious zone beginning at the outlet of the Thermalito Afterbay. This zone is expanding downriver past the confluence of the Yuba River. Adult carcasses produce billions of myxospores annually that move downriver over the winter. C. shasta infection is one of several factors (predation, limited rearing habitat, elevated water temperature, water withdrawal, etc.) limiting natural Chinook Salmon recruitment in the Feather River.
Key words: Ceratonova shasta, Chinook Salmon, Feather River, infectious zone, myxospore
| Citation: J. S. Foott, J. Kindopp, K. Gordon, A. Imrie, and K. Hikey. 2023. California Fish and Wildlife Journal 109:e9. |
| Editor: Steve Parmenter, Inland Deserts Region (retired) |
| Submitted: 30 September 2022; Accepted: 13 March 2023 |
| Copyright: ©2023, Foott et al. This is an open access article and is considered public domain. Users have the right to read, download, copy, distribute, print, search, or link to the full texts of articles in this journal, crawl them for indexing, pass them as data to software, or use them for any other lawful purpose, provided the authors and the California Department of Fish and Wildlife are acknowledged. |
| Funding: Partial funding for this work was obtained from California Department of Water Resources Contract 4600013554. |
| Competing Interests: The authors have not declared any competing interests. |
Introduction
The lower Feather River is a tributary of the Sacramento River in Northern California and divided by flow regime into two distinct reaches (Fig. 1). The low flow channel (LFC) is a partially bypassed reach of the natural channel which extends from Oroville dam (river kilometer (rkm) 117.5) to the Thermalito Afterbay Outlet (TAO, rkm 94.9), a LFC spawning reach (rkm 96.5 – 107.8), and a high flow channel (HFC) beginning below the outlet to the confluence with the Sacramento River. Most of Lake Oroville’s water is diverted through the Thermalito Forebay complex with approximately 25% or less of annual flow released down the LFC. The Thermalito complex warms cold reservoir water for agriculture and additional storage (Seesholtz et al. 2004). Water not diverted for agriculture is returned to the lower Feather River via the TAO. The HFC receives planktonic organisms flushed from the mesotrophic Thermalito complex (Seesholtz et al. 2004).

Both fall and spring-run Chinook salmon (Oncorhynchus tshawytscha) are endemic to the lower Feather River with ≥ 95% of adult spawning occurring in the LFC (Sommer et al. 2001). Since 2008, Feather River Fish Hatchery returns dominate the adult spawning population (Willmes et al. 2018). Emigration of juveniles occurs primarily as fry (< 50 mm) beginning in December and peaking in the January – February period (Sommer et al. 2001; Seeshotz et al. 2004). Despite natural spawning populations averaging 60,000 adults, natural recruitment is deemed quite low. Mercer and Kurth (2014) report a range of < 1% to 10% natural-spawner contribution to the hatchery between 2009–2011.
Ceratonova (syn. Ceratomyxa) shasta Noble 1950 (Atkinson et al. 2014) infects freshwater salmonid fishes and is enzootic to anadromous fish tributaries of the Pacific Northwest of the USA, including the Feather River (Hendrickson et al. 1989). Infection by C. shasta often results in enteronecrosis and anemia in juvenile salmon. This disease is a significant mortality factor for juvenile Chinook salmon in the Klamath River (Foott et al. 2004; Stocking et al. 2006; Fujiwara et al. 2011; Hallett et al. 2012). C. shasta has a complex life cycle, involving an invertebrate polychaete host (Manayunkia occidentalis)as well as the vertebrate salmon host (Atkinson et al. 2020; Bartholomew et al. 1997). Infected polychaetes release actinospores into the water where they attach to the salmon’s gill epithelium, invade into the blood, replicate, and later migrate to the intestinal tract for further multiplication and sporogony (Bjork and Bartholomew 2010). Depending on actinospore genotype and density, innate host resistance, and water temperature, infected fish can develop varying degrees of enteritis and associated anemia (Udey et al. 1975; Foott et al. 2004; Bjork and Bartholomew 2009; Ray et al. 2010; Hallett et al. 2012).
After approximately two weeks post-infection, the parasite can form the myxospore stage. Myxospore release typically occurs after the fish dies. If ingested by the filter-feeding polychaete it completes the life cycle after invading the worm’s gut epithelium (Meaders and Hendrickson 2009). M. occidentalis inhabits tubes constructed of fine sediment and mucus in a variety of lotic conditions with highest densities found in low velocity areas (Stocking and Bartholomew 2007). It likely has an annual generation time (Willson et al. 2010). The myxosporean, Parvicapsula minibicornis, shares the same hosts and range as C. shasta (Bartholomew et al. 2006). It has a tropism for the kidney nephron and can induce glomerulonephritis in the salmon host. It is often a co-infection with C. shasta with 30–52% prevalence of infection in juvenile Feather River salmon during the 2015–2020 study period (Foott et al. 2004). While glomerulonephritis is an additive stressor, hemorrhagic enteronecrosis associated with C. shasta is the primary mortality factor for juvenile salmon. This report describes trends of C. shasta infection in lower Feather River natural Chinook juveniles and the water borne spore stages of the parasite.
Methods
Natural Chinook Salmon juveniles collected by California Department of Water Resource (CDWR) biologists from either rotary screw traps or beach seine (under California Department of Fish and Wildlife Scientific Collection Permit 9611 and 13341). Collections occurred from 2015–2020; however, no data are presented for 2017. Extreme high flows in 2017 reduced sample numbers to approximately 30% of our target (LFC = 84 and HFC = 315 over the 15-week period), limited actual collection period by half, and changed collection methods from rotary screw traps to limited beach seine collections. Staff transported natural fry (prognosis study) or hatchery “cage exposure” fish (sentinel) to the California–Nevada Fish Health Center wet lab and reared in 370 L circular tanks with aeration and flow-through, ambient river temperature water with daily feeding. Fish rearing and euthanization followed published guidelines (Jenkins et al. 2014). Dates reported as Julian Week (JW; Table 1). In 2018, staff transported eight groups of “prognosis” fry from the Herringer trap to the wet laboratory and reared at ambient river temperatures for 21 days. This action occurred on a bi-weekly basis beginning JW5 through JW19 (due to high C. shasta mortality, the last group sampled after only 6 days). We used juvenile fall-run Chinook Salmon from Coleman National Fish Hatchery (Anderson, CA, USA) and Feather River Fish Hatchery (Oroville, CA, USA) in sentinel fish trials in 2015, 2016, and 2019. These populations are historically negative for C. shasta infection and tested negative prior to their exposures. Sentinel trials provide data on infection response of salmon to known exposure durations at specific river locations and dates. We exposed sentinels in the river within 0.01 m3 live cages, transported and reared in the wet lab for 17 to 21 days post-exposure, and sampled for histology. Intestines from mortalities assayed for C. shasta DNA by Quantitative Polymerase Chain Reaction (QPCR). Water temperature for sentinel exposures and the Gridley site measured by Onset probes (Onset, Bourne, MA, USA, https://www.onsetcomp.com).
Table 1. Julian week and corresponding calendar dates.
| Julian Week | Inclusive Dates |
| 1 | 01 Jan–07 Jan |
| 2 | 08 Jan–14 Jan |
| 3 | 15 Jan–21 Jan |
| 4 | 22 Jan–28 Jan |
| 5 | 29 Jan– 04 Feb |
| 6 | 05 Feb–11 Feb |
| 7 | 12 Feb–18 Feb |
| 8 | 19 Feb–25 Feb |
| 9 | 26 Feb–04 Mar |
| 10 | 05 Mar–11 Mar |
| 11 | 12 Mar–18 Mar |
| 12 | 19 Mar–25 Mar |
| 13 | 26 Mar–01 Apr |
| 14 | 02 Apr–08 Apr |
| 15 | 09 Apr–15 Apr |
| 16 | 16 Apr–22 Apr |
| 17 | 23 Apr–29 Apr |
| 18 | 30 Apr–06 May |
Histology
We fixed euthanized fish in Davidson’s fixative for histological examination or froze for QPCR analysis. Kidney, gill, and gastrointestinal tract were processed for 5μm paraffin sections and stained with hematoxylin and eosin (Humason 1979). Fry, smaller than 50mm, were de-calcified and processed as sagittal sections. We placed all tissues for a given fish on one slide and identified by a unique number code. Histological rankings of C. shasta-2 (CS2) ‘clinical disease’ were based on the presence of multifocal lesions of lamina propria hyperplasia or necrosis associated with parasite infection. The CS1 rating required the presence of the parasite but with minimal inflammatory changes.
Quantitative Polymerase Chain Reaction (QPCR)
We collected gill tissue (all arches) and intestine (rectum to the junction of small intestine with the pyloric ceca) separately and extracted DNA with Applied Biosystems MagMax express-96 Magnetic Particle Processer. A C. shasta QPCR assay targeting the 18S ribosomal DNA sequence was used to assay DNA extracted from fish tissues: forward primer (Cs-1034F 5’ CCA GCT TGA GAT TAG CTC GGT AA), reverse primer (Cs-1104R CCC CGG AAC CCG AAA G), and probe (CsProbe-1058T 6FAMCGA GCC AAG TTG GTC TCT CCG TGA AAA C TAMRA) (Hallett and Bartholomew 2006). We performed all reactions (30µL) in a 96-well optical reaction plate using 0.9 µM of both primers, 0.25 µM of probe, 15 µL of TaqMan Universal PCR Master Mix, and 5uL of DNA template. Samples were tested in single well, and reactions were assayed using a 7300 Sequence Detection System (Applied Biosystems) with the following cycling conditions: 2 min at 50°C and 10 min at 95°C, followed by 40 cycles of 95°C for 15 sec and 60°C for 1 min. Each assay plate included a standard curve with three concentrations of reference standards (two replicates each) at known DNA copy number and two negative control wells. We obtained the C. shasta reference standard curve using synthesized DNA (gBlock Gene Fragments, Integrated DNA Technology, Coralville Iowa) containing the 18S ribosomal DNA target sequence. Specifically, 1 ng of DNA, corresponding to 6.83×109 copies of C. shasta DNA was serially diluted over ten orders of magnitude in Tris-ethylenediaminetetraacetic acid buffer. Using QPCR analysis software, we calculated the cycle threshold (CT) values for each standard concentration (SDS software 7300 SDS v 1.4, Applied Biosystems). Criteria for a positive test result required samples to produce a change in normalized fluorescent signal (Δ Rn) greater than or equal to 100,000 florescent units and ≥ 5 copies. Low copy numbers are not reliable given the limitations of the Poisson distribution (Applied Biosystems 2016). A “disease” threshold of > 3.0 logs of C. shasta DNA were used to segregate data based on the relationship between this value and histological lesion of the intestine (Voss et al. 2020).
Water-borne Spore Measurement
We collected water samples with two methods: 1) four 1-liter simultaneous grab samples (majority of “single day longitudinal” water samples), or 2) four 1-liter samples obtained from a 24 h composite sample at the Herringer trap (Global Water Sampler WS700, ~600 mL each hour). Sample sites for both fish and water collection identified in Figure 1. Filtered (47mm, 5µm nitrocellulose, Millipore) water samples were analyzed for C. shasta DNA by Oregon State University (Hallett et al. 2012) and reported as mean spore/L for a one-day site collection.
Myxospores in Carcass and Water
In 2014, 2015, and 2020, we collected intestines from both fall and spring-run Chinook adult carcasses in the spawning reach (rkm 96.5–107.8). A scraping of the intestinal epithelium and lamina propria layers were examined microscopically and myxospores enumerated per mass of scraping material (Foott et al. 2016). Water sampling from the spawning reach and immediately above TAO (Eye trap rkm 96) conducted by three different methods to estimate myxospore concentration post-spawning. All filter extraction and QPCR analysis were conducted by Dr. Stephen Atkinson’s laboratory at Oregon State University. In 2015, triplicate 1-L grab samples analyzed for C. shasta DNA as described above (Hallett et al. 2012). In 2016, 15-L samples were collected at the same locations, suspended particles allowed to settle for 24 hours, slow siphoning of the water column to the 1-L mark, and filtering of the lower 1-L suspension for C. shasta DNA analysis as described above. Spore/L estimates are divided by 15 to obtain concentration data. In 2020, 15-L samples passed through a 142 mm, 5.0 µm nitrocellulose filter (Merck Millipore Ltd) held in an in-line filter holder (Geotech Environmental Equipment, Inc.) under vacuum and the filter was processed in similar fashion as done for water samples.
Data Analysis
We performed linear regression and t-tests with SigmaPlot 12 software (Systat software Inc., San Jose, CA, USA) and calculated 95% confidence limit as standard error of the mean (SEM = standard deviation/squared root of sample number) multiplied by 1.96.
Results
Juvenile Salmon Infection
Prevalence of infection and severity.—Limited histological examination of Chinook juveniles in April and May 2012–2014 demonstrated a marked difference in prevalence of C. shasta infection between LFC (0–5%) and HFC (≥ 68%) (USFWS National Wild Fish Health Survey, www.fws.gov/project/national-wild-fish-health-survey-mapper, June 2023). This disparity continued in the 2015–2020 surveys (Table 2). The more sensitive QPCR assay detected a range of 45–72% prevalence of infection in HFC salmon compared to 9–38% from LFC. Weekly prevalence data shows a marked increase in both infection and severity between Julian weeks (JW) 9–12 (Fig. 2). Prevalence of histological rating of CS2 (clinical disease) was similar to QPCR samples with ≥ 3 logs of parasite DNA in 2018, 2019, and 2020 (Fig. 2). Despite high flows in 2017 and relatively low spore/L concentrations (≤ 25) in 2018, most fish sampled in 2018 were rated as diseased (Table 2).
Table 2. Prevalence of Ceratonova shasta infection (± 95% CI) in juvenile Chinook Salmon intestine samples collected in the low or high flow reaches of the lower Feather River. Collections occurred from 1 Jan–30 Apr from 2015–2020. Data from fish assayed by histology or quantitative polymerase chain reaction (QPCR).
| Year | High Flow Histology | High Flow QPCR | Low Flow Histology | Low Flow QPCR |
| 2015 | 58% ± 12% (n = 64) | 72% ± 10% (n = 71) | 0% (n = 60) | 9% ± 6% (n = 70) |
| 2016 | 46% ± 9%, (n = 106) | 61% ± 11% n = 64) | 0% (n = 15) | 28% ± 15% (n = 36) |
| 2018 | 40% ± 8% (n = 149) | 58% ± 8% (n = 154) | 0% (n = 32) | 38% ± 16% (n = 37) |
| 2019 | 34% ± 8% (n = 128) | 45% ± 8% (n = 164) | 18% ± 12% (n = 38) | 32% ± 14% (n = 44) |
| 2020 | 48% ± 8% (n = 159) | 56% ± 8% (n = 164) | 2% ± 4% (n = 46) | 19% ± 11% (n = 53) |
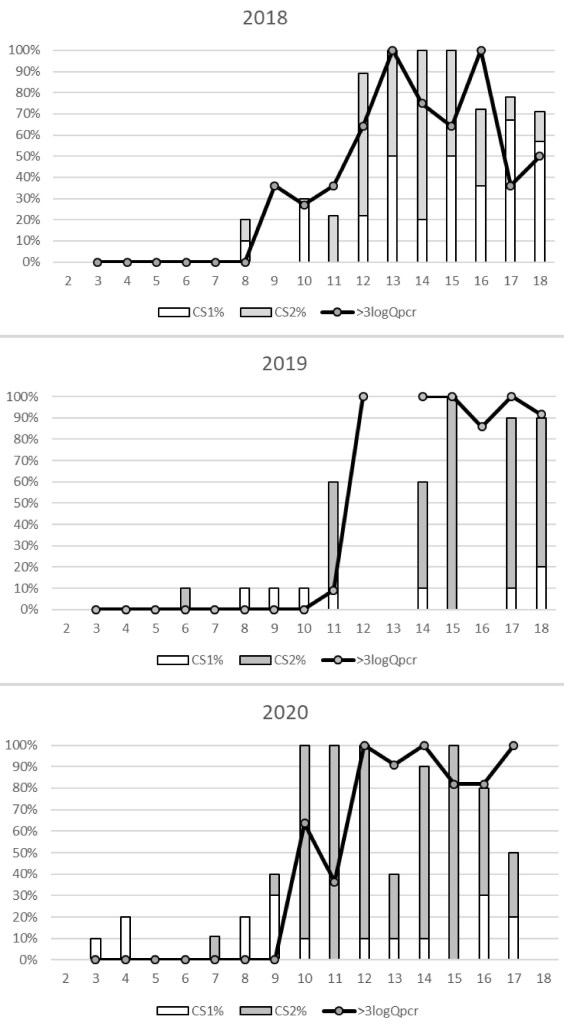
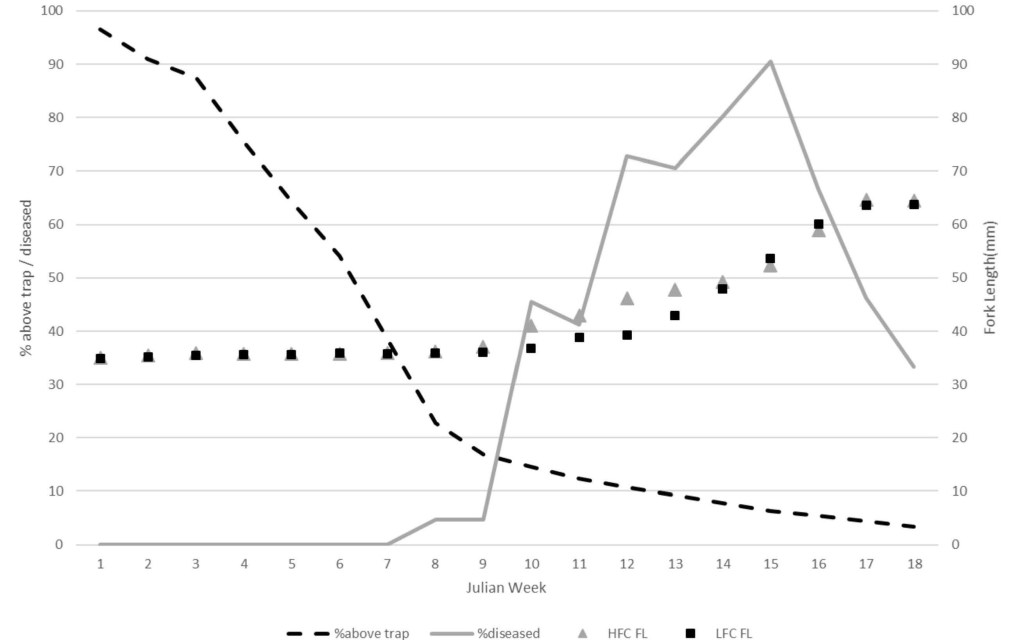
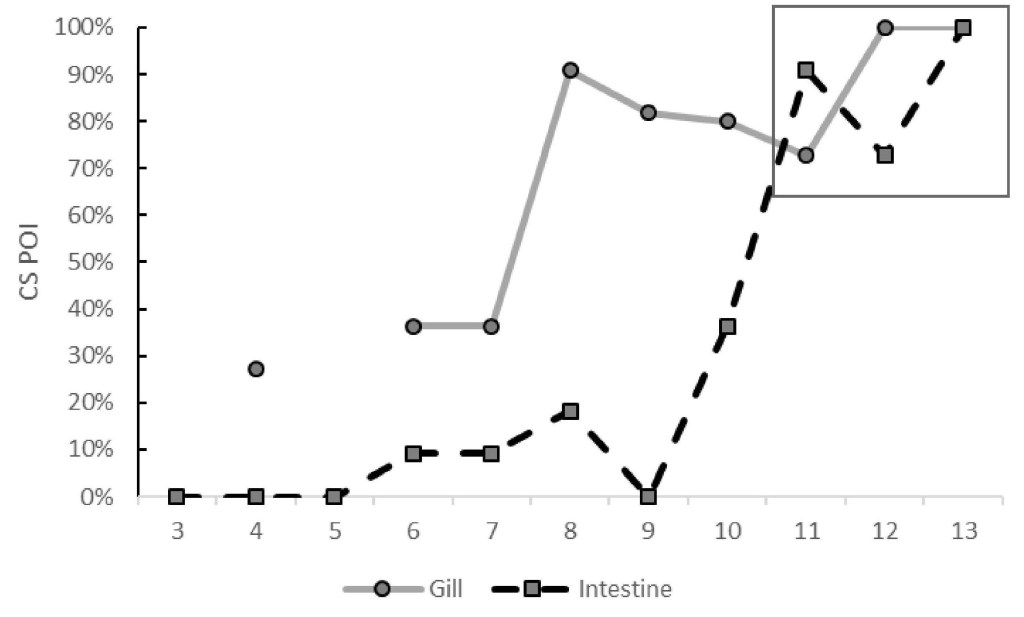
Ceratonova shasta-associated mortality in the 2018 prognosis fry ranged from 0 to 85% with a peak in JW12. Disease prevalence in 21-day survivors mirrored the mortality with the peak (100%) in JW 12. Fry collected at Herringer trap were larger compared to Eye trap cohorts captured during JW10-13 (Fig. 3). This period corresponded with marked increase in the number of disease- rated fry at the HFC trap (Fig. 3). For instance, between JW10-13 of 2019 there was good correlation (R2 of 0.506) between fork length and parasite copy number in Herringer trap fry (Fork length = 56.972 – (0.512 *Copy number), df = 52, P < 0.001). JW10 is also when more than 85% of natural fry have migrated past the Herringer trap (five-year average, Fig 3). 2015 was an outlier to this pattern as the increase in diseased fry (55%) occurred in JW8. In 2020, both gill and intestine were assayed by QPCR. We detected infection much earlier in gill tissue (JW 3 compared to JW6 from intestine) and at greater prevalence for weeks prior to any increase in intestinal infections (Fig. 4).
Sentinel salmon infection.—In March and April of 2015 2016, and 2019, uninfected hatchery salmon were exposed to the lower Feather River for durations of 3–11 days (Table 3). These exposures showed the effect of exposure site, date, and duration on both infection and progression to a diseased state. In 2015, a 3-day exposure at two sites within the presumptive infectious zone showed both prevalence of infection and disease severity was greater at the downriver site (rkm 83.7) compared to rkm 93 (Table 3). A 6-day exposure at rkm 80.5 resulted in a similar prevalence of infection and disease as the 3-day exposure at rkm 83.7 however, it was much higher (70% prevalence of infection) than a similar 6-day exposure at rkm 35.9 (17%). The 11-day exposure during JW 15 showed a similar pattern with all fish from the rkm 80.5 site experiencing clinical disease compared to only 7% diseased at the lower river (rkm 29) site (Table 3). In 2016, three- or four-day exposures at rkm 80.5 had similar prevalence of infection (78 and 82%) and moderate levels of disease (7 and 13%) when conducted in February (JW 7) and late March (JW 13). Actinospore concentrations were deemed high (140 spores/L) for both exposures. In 2019, two- and four-day exposures at rkm 93, during February and March, were conducted to simulate typical fry transient times between LFC to the HFC trap site. Water samples at the exposure site measured 1–20 spores/L. The 4-day exposure groups had 2.3–6.4 greater POI (32–80%) than the 2-day groups (5–15%). None of the sentinels showed signs of disease.
Table 3. Prevalence (and %) of Ceratonova shasta infection (CS-POI) and fish rated as diseased from sentinel salmon exposed to the lower Feather River in Julian week (JW) 7–15 during 2015, 2016, and 2019. For each exposure, sites recorded in river kilometer (rkm), duration as days of exposure (DE), and mean daily temperature (MDT, °C).
| Year | Rkm | JW | DE | MDT | CS-POI | Disease |
| 2015 | 93.0 | 12 | 3 | 14 | 14/32 (44%) | 2/32 (6%) |
| 2015 | 93.0 | 15 | 3 | 16 | 14/32 (44%) | 2/32 (6%) |
| 2015 | 83.7 | 12 | 3 | 14 | 20/30 (67%) | 2/30 (7%) |
| 2015 | 83.7 | 15 | 3 | 15 | 24/30 (80%) | 8/30 (27%) |
| 2015 | 80.5 | 12 | 6 | 17 | 14/20 (70%) | 4/20 (20%) |
| 2015 | 80.5 | 15 | 11 | 16 | 15/15 (100%) | 15/15 (100%) |
| 2015 | 35.9 | 12 | 6 | 17 | 3/18 (17%) | 0/18 (0%) |
| 2015 | 29.0 | 15 | 11 | 18 | 15/28 (18%) | 2/18(7%) |
| 2016 | 80.5 | 7 | 3 | 12 | 35/45 (78%) | 6/45 (13%) |
| 2016 | 80.5 | 13 | 4 | 15 | 24/28 (82%) | 2/28 (7%) |
| 2019 | 93.0 | 7 | 2 | 9 | 1/20 (5%) | 0/20 (0%) |
| 2019 | 93.0 | 7 | 4 | 9 | 6/20 (32%) | 0/20 (0%) |
| 2019 | 93.0 | 9 | 2 | 10 | 3/21 (14%) | 0/21 (0%) |
| 2019 | 93.0 | 9 | 4 | 10 | 16/20 (80%) | 0/20 (0%) |
| 2019 | 93.0 | 11 | 2 | 10 | 3/20 (15%) | 0/20 (0%) |
| 2019 | 93.0 | 11 | 4 | 10 | 7/20 (35%) | 0/20 (0%) |
Spore Stages
Myxospore monitoring.—Prevalence of myxospores from adult carcasses was 53% (32/60), 80% (48/60), and 70% (14/20) in 2014, 2015, and 2020, respectively. The estimated quantity of myxospores observed in the intestine samples ranged from 49–875 million. We consider samples with 500,000 myxospores/scraping or more to be the major spore contributors to the river. These high spore contributors ranged from 21% (2020 samples) to 65% (2015). In 2015, two to three spores/L were measured in spawning reach (rkm 104.7) water samples between October and December (Table 4). In contrast, C. shasta DNA (2-5 spores/L) was only detected at Eye trap (directly above HFC) in October and early November (Table 4). A positive signal (> 0.03 spore/L) detected from both the spawning reach and Eye trap sites from late October 2016 through early January 2017. Heavy rains in the winter of 2017 increased turbidity and PCR inhibition occurred in 60% of the water samples. In 2020, positive signal was detected at Eye trap from early December through the last sample in early February. The spawning reach sample was only positive in early December. In contrast to the low spore concentrations discussed above, water samples within the HFC are >10 spores/L by mid-January. Fry infection currently indicates the presence of fish-infective actinospores. Spore concentration was markedly higher at these two sites in 2015 compared to 2016–2017 and 2020–2021. QPCR assay of filtered water cannot distinguish between fish-infective actinospores and worm-infective myxospores.
Table 4. Presumptive myxospore concentration (spore/L) in water samples collected from the spawning reach (rkm 104.7) and directly above the high flow channel (Eye trap, rkm 96.5). Sample collection occurred during and after 2015, 2016, and 2020 Chinook spawning (Oct–Feb).
| Date | rkm 104.7 | rkm 96.5 |
| 15 Oct 2015 | 1.72 | 1.26 |
| 21 Oct 2015 | 2.63 | 4.15 |
| 4 Nov 2015 | 2.77 | 2.13 |
| 20 Nov 2015 | 1.73 | 0 |
| 10 Dec 2015 | 2.93 | 0 |
| 27 Oct 2016 | 0.19 | 0.05 |
| 3 Nov 2016 | 0.39 | 0 |
| 9 Nov 2016 | 0.51 | 0.14 |
| 16 Nov 2016 | 0.26 | 0.03 |
| 1 Dec 2016 | 0.03 | 0.04 |
| 7 Dec 2016 | 0.12 | 0.09 |
| 15 Dec 2016 | 0.05 | 0.03 |
| 21 Dec 2016 | 0 | 0 |
| 28 Dec 2016 | 0.08 | 0.03 |
| 5 Jan 2017 | 0 | 0 |
| 2 Dec 2020 | 1.33 | 2.29 |
| 16 Dec 2020 | 0 | 0.48 |
| 31 Dec 2020 | 0.13 | 0.86 |
| 13 Jan 2021 | 0.28 | 0.67 |
| 28 Jan 2021 | 0.1 | 0.76 |
| 10 Feb 2021 | 0 | 0.57 |
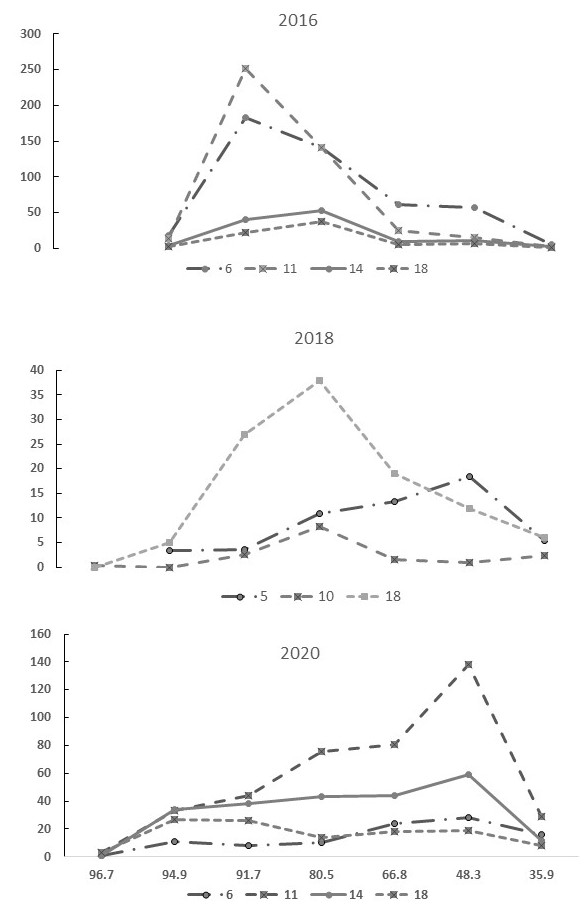
HFC spore concentration.—Herringer trap spore/L data provides insight into conditions within the infectious zone (Fig. 5). Except for 2018, peak spore concentrations (≥ 120 spores/L) tend to occur between JW 7–10 (Table 5). Initial detection of C. shasta infection in Chinook fry occurred 1–7 weeks prior to the peak spore concentration and 0–4 weeks after spore concentrations reached ≥ 10 spores/L (Table 5). Mean daily water temperature was 9.5–13.2°C during these annual peaks. A series of samples, referred to as longitudinal sampling (collected on one day in an upstream manner at multiple locations), demonstrate spatial and temporal differences in actinospore concentration within and between years (Table 6, Fig. 6). Timing of peak concentration biased by irregular sample collection schedule but tended to be in JW 11. Peak spore concentration was earlier (JW6) in 2019 and later (JW18) in 2018 (Table 6). 2018 was also unique in the markedly lower spore concentrations measured (peak was 4.8 times lower than 2019 peak). Longitudinal profiles demonstrate the markedly higher spore concentrations within an “infectious zone” downstream of TAO (rkm 94.9, Fig. 6). The location of peak spore concentration shifted downriver starting in 2019 from rkm 91.7–80.5 to rkm 73.7–49.1. Two lines of evidence suggest that the afterbay is not the primary source of actinospore production but rather the high flow reach itself. In May 2015, water samples collected directly above the TAO in the LFC (4.2 spores/L), within the afterbay channel (0.3 spores/L, outlet – river mixing zone (1.7 spores/L), and one rkm downriver of outlet (12.9 spores/L) indicate the minimal contribution of the afterbay. Similarly, no parasite DNA was detected in samples from the channel in January and February 2019.

Table 5. Herringer trap Ceratonova shasta occurrence (spore/L) with temperature (°C) and Julian week (JW) for peak spore concentration and Julian week of initial detection of infection and greater than 10 sp/L in Chinook Salmon fry.
| Year | JW (peak sp/L) | °C | sp/L | JW (≥ 10 sp/L) | JW (first infection) |
| 2016 | 8 | 12.1 | 168 | 1 | 4 |
| 2018 | 12 | 10.9 | 25 | 4 | 8 |
| 2019 | 7 | 9.5 | 183 | 4 | 5 |
| 2020 | 10 | 13.2 | 120 | 3 | 3 |
Table 6. Ceratonova shasta spore/L longitudinal data collected from rkm 96.7 to 35.9 during Julian weeks (JW) 5–18 of 2015, 2016, 2018, 2019, and 2020. Peak spore/L for a given year is in bold type. (NT = not taken)
| Year | JW | rkm 96.7 | rkm 94.9 | rkm 91.7 | rkm 80.5 | rkm 73.7 | rkm 66.8 | rkm 49.1 | rkm 35.9 |
|---|---|---|---|---|---|---|---|---|---|
| 2015 | 11 | NT | 14 | 294 | 175 | NT | NT | 11 | 1 |
| 2016 | 6 | NT | 18 | 183 | 141 | NT | 62 | 57 | 6 |
| 2016 | 11 | NT | 14 | 252 | 141 | NT | 25 | 15 | 2 |
| 2016 | 14 | NT | 5 | 41 | 53 | NT | 9 | 12 | 2 |
| 2016 | 18 | NT | 2 | 22 | 38 | NT | 6 | 7 | 1 |
| 2018 | 5 | NT | 3 | 4 | 11 | NT | 18 | 13 | 5 |
| 2018 | 10 | 0 | 0 | 3 | 8 | NT | 2 | 1 | 2 |
| 2018 | 18 | 0 | 5 | 27 | 38 | NT | 19 | 12 | 6 |
| 2019 | 5 | 0 | NT | 37 | NT | 50 | NT | 27 | 15 |
| 2019 | 6 | 1 | 0 | 115 | NT | 183 | 103 | 103 | 4 |
| 2019 | 8 | 0 | NT | NT | 107 | 128 | NT | 82 | 2 |
| 2019 | 10 | NT | NT | NT | NT | 21 | 8 | 16 | 7 |
| 2019 | 11 | NT | NT | 3 | NT | 22 | 3 | NT | 11 |
| 2019 | 16 | 0 | NT | 5 | NT | 3 | 5 | 6 | 5 |
| 2020 | 6 | 1 | 11 | 8 | 10 | NT | 24 | 28 | 16 |
| 2020 | 11 | 3 | 33 | 44 | 76 | NT | 81 | 138 | 29 |
| 2020 | 14 | 1 | 34 | 38 | 43 | NT | 44 | 59 | 12 |
| 2020 | 18 | 3 | 27 | 26 | 14 | NT | 18 | 19 | 8 |
Discussion
Pathogen infection and disease are associated with declining Pacific salmon populations and are a concern for Central Valley Chinook stocks (Foott et al. 2007; Fujiwara et al. 2011; Furey et al. 2020; Lehman et al. 2020). The relationship between C. shasta infection of juveniles and the observed low recruitment of natural Feather River salmon (< 10 % of adult returns) is undefined currently. Ceratomyxosis is likely a significant factor in juvenile Feather River Chinook salmon survival. We observed prevalence of infection averaging 45% (histology) and 58% (QPCR) in natural fry within the HFC over the five-year period. Initial fry infection occur as actinospore concentration increases during late January through early February. By March over 50 % of infected fry rated as diseased with little probability of survival. Conversely, ≥ 85% of the natural fry population have migrated past the Herringer trapping site when disease severity increases in March. Determination of a fish’s infectious status, from its intestine sample, is problematic as 7- 10 days post-infection is required for the parasite to move through the circulatory system and invade the intestinal tract (Bjork and Bartholomew 2010). Once fry leave the LFC, they tend to move rapidly within the HFC and pass the trap site in a week or less (Seesholtz et al. 2004, CDWR mark-recapture trap data). Our sentinel studies demonstrate that disease progression occurs if exposure within the HFC is 5 days or more. The observation of diseased fry at Herringer trap in March corresponds with the capture of larger fish (≥ 40 mm FL) suggesting they rear for several weeks within the HFC prior to capture. Disease mortality of larger fry reduces this life history “portfolio” for Feather River natural Chinook. The use of intestine samples alone will underestimate the prevalence of infection in migrant fry. Gill infection in these early out-migrants suggests some proportion will progress into a disease state. This hypothesis is also based on the wide area of infectivity and hence extended exposure fry experience in the Feather River. Survey efforts downriver of Herringer trap may elucidate the question of disease progression in fry chronically exposed to low actinospore concentrations as they migrate downriver in January and February.
An infectious zone, beginning from the TAO (rkm 94.9), has enlarged from the 2015 survey to include reaches below Yuba River confluence. It is unclear if polychaete density has also increased downriver since 2015. An understanding of the polychaete distribution, locations of protected habitats, and seasonal density would inform management decisions for this disease issue. Alexander et al. (2014) reported a modeling approach in predicting polychaete abundance and response to flow conditions. The high flows of 2017, followed by increasing levels of infection in 2018 and 2019, suggest that some portion of the polychaete population is associated with stable habitat such as lee side of boulders or riprap and can re-colonize within a year. Other workers have reported similar occurrences following disturbances (Malakauskas et al. 2013; Alexander et al. 2014). Another ecological question is why the TAO is a consistent feature of the upriver boundary for the infectious zone. Planktonic food input may be a factor, however; other environmental and habitat features could also be important (Seesholtz et al. 2004; Alexander et al. 2014). A comparison of LFC to HFC habitat characteristics is needed for this analysis.
Myxospore dispersion from adult salmon carcasses is another avenue of potential management. Adult carcasses produce billions of myxospores each year that are later ingested by the alternate worm host, Manayunkia occidentalis, to continue the parasite’s life cycle (Bartholomew et al. 1997; Atkinson et al. 2020). Adult carcasses produce the vast majority of myxospores that feed into the annual polychaete infection cycle (Foott et al. 2016; Kent et al. 2014; Atkinson et al. 2020). Reducing myxospore transmission to polychaetes will require an understanding of flow magnitude, duration, and seasonal timing to optimize the movement of myxospores past the current infectious zone. This study described a parasitic disease that results in high mortality for a portion of the juvenile Chinook population in the lower Feather River. This disease has a defined zone of infectivity beginning below the TAO and a seasonality that begins in late January or early February.
Acknowledgments
The findings and conclusions of this article are those of the authors and do not necessarily represent the views of the U.S. Fish and Wildlife Service or California Department of Water Resources. Partial funding for this work was obtained from California Department of Water Resources Contract 4600013554. Laboratory assay assistance provided by Fish Health Center staff S. Freund, A. Voss, R. Stone, and J. Jacobs. Some field collections conducted by the Department of Water Resources’ biologist M. Barreras. Water sample analysis by Dr. S. Atkinson, Oregon State University.
Literature Cited
- Alexander, J. A., S. L. Hallett, R. W. Stocking, L. Xue, and J. L. Bartholomew. 2014. Host and parasite populations after a ten-year flood: Manayunkia speciosa and Ceratonova (syn Ceratomyxa) shasta in the Klamath River. Northwest Science 88:219–233.
- Applied Biosystems. 2016. Application Note: Real-time PCR: Understanding Ct. Publication CO019879 0116. Available from: https://www.thermofisher.com/content/dam/LifeTech/Documents/PDFs/PG1503-PJ9169-CO019879-Re-brand-Real-Time-PCR-Understanding-Ct-Value-Americas-FHR.pdf
- Atkinson, S. D., J. L. Bartholomew, and G. W. Rouse. 2020. The invertebrate host of salmonid fish parasites Ceratonova shasta and Parvicapsula minibicornis (Cnidaria:Myxozoa), is a novel fabriciid annelid, Manayunkia occidentalis sp. nov. (Sabellida: Fabriciidae). Zootaxa 4751:310–320.
- Atkinson, S. D., J. S. Foott, and J. L. Bartholomew. 2014. Erection of Ceratonova N. gen. (Myxosporea: Ceratomyxidae) to encompass histozoic species C. gasterostea N. Sp. from threespine stickleback (Gasterosteus aculeatus) and C. shasta N. comb. from salmonid fishes. Journal of Parasitology 100(5):640–645.
- Bartholomew, J. L., S. D. Atkinson, and S. L. Hallett. 2006. Involvement of Manayunkia speciosa (Annelida: Polychaeta: Sabellidae) in the life cycle of Parvicapsula minibicornis, a myxozoan parasite of Pacific salmon. Journal of Parasitology 92:742–748.
- Bartholomew, J. L., M. Whipple, D. Stevens, and J. Fryer, 1997. The life cycle of Ceratomyxa shasta, a myxosporean parasite of salmonids, requires a freshwater polychaete as an alternate host. Journal of Parasitology 83:859–868.
- Bjork, S. J., and J. L. Bartholomew. 2010. Invasion of Ceratomyxa shasta (Myxozoa) and comparison of migration to the intestine between susceptible and resistant fish host. International Journal of Parasitology 40:1087–1095.
- Bjork, S. J., and J. L. Bartholomew. 2009. Effects of Ceratomyxa shasta dose on a susceptible strain of rainbow trout and comparatively resistant Chinook and Coho Salmon. Diseases of Aquatic Organisms 86:29–37.
- Foott, J. S., R. Stone, R. Fogerty, K. True, A. Bolick, J. L. Bartholomew, S. L. Hallett, G. R. Buckles, and J. D. Alexander. 2016. Production of Ceratonova shasta myxospores from salmon carcasses: carcass removal is not a viable management option. Journal of Aquatic Animal Health 28:75–84.
- Foott, J. S., R. Stone, and K. Nichols. 2007. Proliferative kidney disease (Tetracapsuloides bryosalmonae) in Merced River hatchery juvenile Chinook Salmon: mortality and performance impairment in 2005 smolts. California Fish and Game 93:57–76.
- Foott, J. S., R. Harmon, and R. Stone. 2004. Effect of water temperature on non-specific immune function and ceratomyxosis in juvenile Chinook Salmon and Steelhead from the Klamath River. California Fish and Game 90:71–90.
- Fujiwara, M., M. S. Mohr, A. Greenberg, J. S. Foott, and J. L. Bartholomew. 2011. Effects of Ceratomyxosis on population dynamics on Klamath Fall-run Chinook Salmon. Transactions of the American Fisheries Society 140:1380–1391.
- Furey, N. B., A. L. Bass, K. M. Miller, S. Li, A. G. Lotto, S. J. Healy, S. M. Drenner, and S. G. Hinch. 2021. Infected juvenile salmon can experience increased predation during freshwater migration. Royal Society Open Science 8:201522.
- Hallett, S. L., R. A. Ray, C. N. Hurst, R. A. Holt, G. R. Buckles, S. D. Atkinson, and J. L. Bartholomew. 2012. Density of the waterborne parasite Ceratomyxa shasta and its biological effects on salmon. Applied and Environmental Microbiology 78:3724–3731.
- Hallett, S. L., and J. L. Bartholomew. 2006. Application of a real-time PCR assay to detect and quantify the myxozoan parasite Ceratomyxa shasta in water samples. Diseases of Aquatic Organisms 71:109–118.
- Hendrickson, G. L, A. Carleton, and D. Manzer. 1989. Geographic and seasonal distribution of the infective stage of Ceratomyxa shasta (Myxozoa) in Northern California. Diseases of Aquatic Organisms 7:165–169.
- Humason, G. L. 1979. Animal Tissue Techniques. 4th edition. W. H. Freeman and Co., San Francisco, CA, USA.
- Jenkins, J. A., J. D. Bowker, J. R. MacMillan, J. G. Nickum, J. D. Rose, P. W. Sorensen, G. W. Whitledge, J. W. Rachlin, B. E. Warkentine, and H. L Bart. 2014. Guidelines for the Use of Fishes in Research. American Fisheries Society, Bethesda, MD, USA.
- Kent, M. L., K. Soderlund, E. Thomann, C. B. Streck, and T. J. Sharpton. 2014. Post-mortem sporulation of Ceratomyxa shasta (Myxozoa) after death in adult Chinook Salmon. Journal of Parasitology 100:679–683.
- Lehman, B. M, R. C. Johnson, M. C. Johnson, O. T. Burgess, R. E. Connon, N. A. Fangue, J. S. Foott, S. L. Hallett, B. Martinez-Lopez, K. M. Miller, M. K. Purcell, N. A. Som, P. V. Donoso, and A. Collins. 2020. Disease in Central Valley salmon: status and lessons from other systems. San Francisco Estuary & Watershed Science 18(3):art2. doi.org/10.15447//sfews.2020v18iss3art2
- Malakauskas, D. M., S. J. Willson, M. A. Wilzbach, and N. A. Som. 2013. Flow variation and substrate type affect dislodgement of the freshwater polychaete, Manayunkia speciosa. Freshwater Science 32:862–873.
- Meaders, M. D., and G. L. Hendrickson. 2009. Chronological development of Ceratomyxa shasta in the polychaete host, Manayunkia speciosa. Journal of Parasitology 95:1397–1407.
- Mercer, M., and R. Kurth. 2014. An evaluation of otolith thermal marking at the Feather River Hatchery, California. San Francisco Estuary & Watershed Science 12(4):art3. doi.org/10.15447/sfews.2014v12iss4art3
- Noble, E. R. 1950. On a myxosporidian (protozoan) parasite of California trout. Journal of Parasitology 36:457–460.
- Ray, R. A., P. A. Rossignol, and J. L. Bartholomew. 2010. Mortality threshold for juvenile Chinook salmon (Oncorhynchus tshawytscha) in an epidemiological model of Ceratomyxa shasta. Diseases of Aquatic Organisms 93:63–70.
- Seesholtz, A., B. J. Cavallo, J. Kindopp, and R. Kurth. 2004. Juvenile fishes of the lower Feather River: distribution, emigration patterns, and associations with environmental variables. American Fisheries Society Symposium 39:141–166.
- Sommer, T., D. McEwan, and R. Brown. 2001. Factors affecting Chinook spawning in the lower Feather River. Pages 269–297 in R. L. Brown, editor. Contributions to the Biology of Central Valley Salmonids. Fish Bulletin 179. California Department of Fish and Game, Sacramento, CA, USA.
- Stocking, R. W., and J. L. Bartholomew. 2007. Distribution and habitat characteristics of Manayunkia speciosa and infection prevalence with the parasite Ceratomyxa shasta in the Oregon-California Klamath River basin. Journal of Parasitology 93:78–88.
- Stocking, R. W., R. A. Holt, J. S. Foott, and J. L. Bartholomew. 2006. Spatial and temporal occurrence of the salmonid parasite Ceratomyxa shasta in the Oregon-California Klamath River Basin. Journal of Aquatic Animal Health 18:194–202.
- Udey, L. R., J. L. Fryer, and K. S. Pilcher. 1975. Relationship of water temperature to Ceratomyxosis in Rainbow Trout (Salmo gairdneri) and Coho Salmon (Oncorhynchus kisutch). Journal of Fisheries Research Board of Canada 32:1545–1551.
- Voss, A., J. S. Foott, and S. Freund. 2020. Myxosporean parasite (Ceratonova shasta and Parvicapsula minibicornis) prevalence of infection in Klamath River basin juvenile Chinook Salmon, March–July 2020. U.S. Fish and Wildlife Service, California–Nevada Fish Health Center, Anderson, CA, USA. Available from: http://www.fws.gov/canvfhc/reports.html
- Willmes, M., J. A. Hubbs, A. M. Sturrock, Z. Bess, L. S. Lewis, J. J. G. Glessner, R. C. Johnson, R. Kurth, and J. Kindopp. 2018. Fishery collapse, recovery, and the cryptic decline of wild salmon on a major California river. Canadian Journal of Fisheries and Aquatic Sciences 75:1836–1848.
- Willson, S. J., M. A. Wilzbach, D. M. Malakauskas, and K. W. Cummings. 2010. Lab rearing of a freshwater polychaete (Manayunkia speciosa, Sabellidae) host for salmon pathogens. Northwest Science 84:182–190.

